13.3
Impact Factor
Theranostics 2020; 10(6):2612-2620. doi:10.7150/thno.42228 This issue Cite
Research Paper
Targeted alpha therapy in a systemic mouse model of prostate cancer - a feasibility study
1. Ahmanson Translational Theranostic Division, Department of Molecular and Medical Pharmacology, David Geffen School of Medicine at University of California, Los Angeles (UCLA), CA, USA.
2. Department of Translational Medicine, Division of Urological Cancers, Skåne University Hospital Malmö, Lund University, Sweden.
3. Department of Surgery, David Geffen School of Medicine at University of California, Los Angeles (UCLA), CA, USA.
*These authors contributed equally to this study.
Abstract
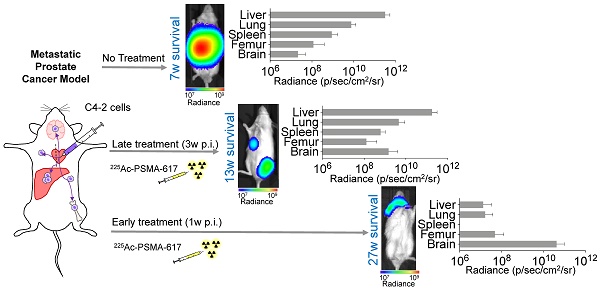
225Ac-PSMA-617 targeted-therapy has demonstrated efficacy in 75-85% of patients; however, responses are not durable. We aimed to establish translatable mouse models of disseminated prostate cancer (PCa) to evaluate effectiveness of 225Ac-PSMA-617 at various disease stages.
Methods: C4-2, C4-2B, or 22Rv1 cells were injected into the left ventricle of male NSG mice. Disease progression was monitored using bioluminescence imaging (BLI). For treatment, mice were injected with 40 kBq 225Ac-PSMA-617 at one (early treatment cohort) or three weeks (late treatment cohort) post-inoculation. Treatment efficacy was monitored by BLI of whole-body tumor burden. Mice were sacrificed based on body conditioning score.
Results: C4-2 cells yielded metastases in liver, lungs, spleen, stomach, bones, and brain - achieving a clinically relevant model of widespread metastatic disease. The disease burden in the early treatment cohort was stable over 27 weeks in 5/9 mice and progressive in 4/9 mice. These mice were sacrificed due to brain metastases. Median survival of the late treatment cohort was superior to controls (13 vs. 7 weeks; p<0.0001) but inferior to that in the early treatment cohort (13 vs. 27 weeks; p<0.001). Late cohort mice succumbed to extensive liver involvement. The 22Rv1 and C4-2B systemic models were not used for treatment due to high kidney metastatic burden or low take rate, respectively.
Conclusion: C4-2 cells reproduced metastatic cancer spread most relevantly. Early treatment with 225Ac-PSMA-617 prevented liver metastases and led to significant survival benefit. Late treatment improved survival without reducing tumor burden in the liver, the main site of metastasis. The current findings suggest that early 225Ac-PSMA-617 intervention is more efficacious in the setting of widespread metastatic PCa.
Keywords: PSMA, targeted alpha therapy, prostate cancer, metastatic mouse model, actinium
 Global reach, higher impact
Global reach, higher impact