Impact Factor
Theranostics 2020; 10(6):2696-2713. doi:10.7150/thno.41001 This issue Cite
Research Paper
Identification of a new aggressive axis driven by ciliogenesis and absence of VDAC1-ΔC in clear cell Renal Cell Carcinoma patients
1. Université Côte d'Azur (UCA), CNRS-UMR 7284-Inserm U1081, IRCAN, Centre Antoine Lacassagne, 33 Ave. de Valombrose, 06189 Nice, France.
2. Present address: Université Côte d'Azur (UCA), INSERM U1065, C3M, 151 Route de St Antoine de Ginestière, BP2 3194, 06204 Nice Cedex 03, France.
3. Medical Biology Department, Centre Scientifique de Monaco (CSM), Monaco.
4. Université Côte d'Azur (UCA), Centre Commun de Microscopie Appliquée, Nice, France.
5. Institut du thorax, INSERM, CNRS, Univ. Nantes, Nantes, France.
6. Ecole Pratique des Hautes Etudes, EPHE, PSL Research University, Paris, France.
7. INSERM UMR 1186, Integrative Tumor Immunology and Genetic Oncology, Gustave Roussy, EPHE, PSL, Fac. de médecine - Univ. Paris-Sud, Université Paris-Saclay, 114 rue Edouard Vaillant, 94800 Villejuif, France.
8. Centre Antoine Lacassagne, Oncology Department, Nice, France.
9. Rennes University, Rennes University Hospital, Department of Pathology, Rennes, France.
10. Centre Hospitalier Universitaire de Nice, Department of Pathology, Nice, France.
11. REDIR Center, Department of Urology, AP-HP, Bicêtre Hospital, 78 Rue du Général Leclerc, 94270 Le Kremlin-Bicêtre.
12. Centre Antoine Lacassagne, Statistics Department, Nice, France.
13. Centre Hospitalier Universitaire de Nice, Department of Vascular Surgery, Nice, France.
14. Université Côte d'Azur (UCA), CNRS-UMR 7370, LP2M, Nice, France.
15. Department of Molecular and Human Genetics, The Mitochondrial Diagnostic Laboratory, Baylor College of Medicine, Houston, TX 77030, USA.
16. Université Côte d'Azur (UCA), CNRS, IPMC, FHUOncoAge, 06560 Valbonne, France.
* These authors contributed equally to this paper.
Abstract
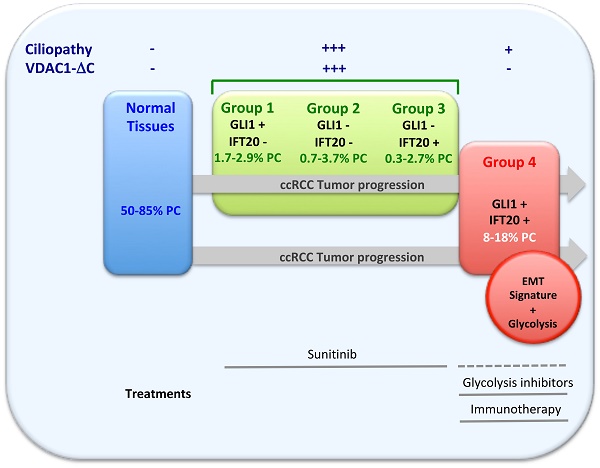
Rationale: Renal cell carcinoma (RCC) accounts for about 2% of all adult cancers, and clear cell RCC (ccRCC) is the most common RCC histologic subtype. A hallmark of ccRCC is the loss of the primary cilium, a cellular antenna that senses a wide variety of signals. Loss of this key organelle in ccRCC is associated with the loss of the von Hippel-Lindau protein (VHL). However, not all mechanisms of ciliopathy have been clearly elucidated.
Methods: By using RCC4 renal cancer cells and patient samples, we examined the regulation of ciliogenesis via the presence or absence of the hypoxic form of the voltage-dependent anion channel (VDAC1-ΔC) and its impact on tumor aggressiveness. Three independent cohorts were analyzed. Cohort A was from PREDIR and included 12 patients with hereditary pVHL mutations and 22 sporadic patients presenting tumors with wild-type pVHL or mutated pVHL; Cohort B included tissue samples from 43 patients with non-metastatic ccRCC who had undergone surgery; and Cohort C was composed of 375 non-metastatic ccRCC tumor samples from The Cancer Genome Atlas (TCGA) and was used for validation. The presence of VDAC1-ΔC and legumain was determined by immunoblot. Transcriptional regulation of IFT20/GLI1 expression was evaluated by qPCR. Ciliogenesis was detected using both mouse anti-acetylated α-tubulin and rabbit polyclonal ARL13B antibodies for immunofluorescence.
Results: Our study defines, for the first time, a group of ccRCC patients in which the hypoxia-cleaved form of VDAC1 (VDAC1-ΔC) induces resorption of the primary cilium in a Hypoxia-Inducible Factor-1 (HIF-1)-dependent manner. An additional novel group, in which the primary cilium is re-expressed or maintained, lacked VDAC1-ΔC yet maintained glycolysis, a signature of epithelial-mesenchymal transition (EMT) and more aggressive tumor progression, but was independent to VHL. Moreover, these patients were less sensitive to sunitinib, the first-line treatment for ccRCC, but were potentially suitable for immunotherapy, as indicated by the immunophenoscore and the presence of PDL1 expression.
Conclusion: This study provides a new way to classify ccRCC patients and proposes potential therapeutic targets linked to metabolism and immunotherapy.
Keywords: Ciliopathy, clear cell Renal Cell Carcinoma, immunotherapy, HIFs, poor prognosis, primary cilium, Voltage-Dependent Anion Channel 1, VDAC1.
 Global reach, higher impact
Global reach, higher impact