Impact Factor
Theranostics 2020; 10(7):3281-3292. doi:10.7150/thno.37599 This issue Cite
Research Paper
EDTA-Modified 17β-Estradiol-Laden Upconversion Nanocomposite for Bone-Targeted Hormone Replacement Therapy for Osteoporosis
1. Department of Geriatrics, Xinhua Hospital of Shanghai Jiaotong University, School of Medicine, Shanghai 200092, China.
2. Department of Chemistry, Fudan University, 220 Handan Road, Shanghai 200433, P.R. China.
3. Shanghai Med-X Engineering Center for Medical Equipment and Technology, School of Biomedical Engineering, Shanghai JiaoTong University, Shanghai 200030, P. R. China
*These authors contributed equally.
Abstract
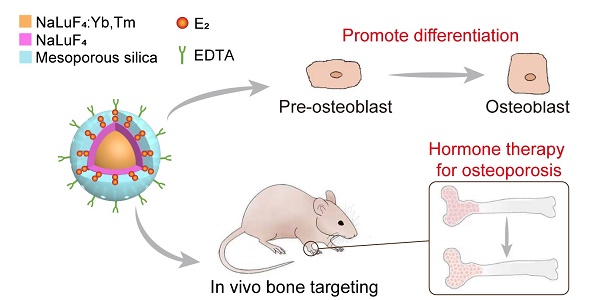
Hormone therapy (HT) is one of the most effective treatments for osteoporosis. However, the nonselective accumulation of hormone in organs such as breast, heart and uterus other than bones causes serious side effects, which impedes the application of HT. Hence, it is critically important to develop a HT strategy with reduced non-specific enrichment of hormone drugs in non-target tissues and enhanced bone-targeting ability.
Methods: Herein, a 17β-estradiol (E2)-laden mesoporous silica-coated upconversion nanoparticle with a surface modification of ethylenediaminetetraacetic acid (EDTA) (NaLuF4:Yb,Tm@NaLuF4@mSiO2-EDTA-E2, E2-csUCNP@MSN-EDTA) is developed for bone-targeted osteoporosis hormone therapy. EDTA was attached onto the surface of E2 upconversion nanocomposite to enhance its affinity and efficiency targeting bone tissue and cells to optimize hormone replacement therapy for osteoporosis. We characterized the size, cytotoxicity, loading and release efficiency, in situ and ex vivo imaging. Further, in vitro and in vivo osteogenic ability was tested using preosteoblast and ovariectomy mouse model of osteoporosis.
Results: The upconversion core of E2-csUCNP@MSN-EDTA nanoparticle serves as an excellent imaging agent for tracking the loaded hormone drug in vivo. The mesoporous silica layer has a high loading efficiency for E2 and provides a relatively long-lasting drug release within 50 h. EDTA anchored on the silica layer endows the nanocomposite with a bone targeting property. The nanocomposite effectively reverses estrogen deficiency-induced osteoporosis and reduces the damage of hormone to the uterus. The bone mineral density in the nanocomposite treatment group is nearly twice that of the ovariectomized (OVX) group. Compared with the E2 group, the uterine weight and luminal epithelial height were significantly lower in the nanocomposite treatment group.
Conclusion: This work demonstrated that E2-csUCNP@MSN-EDTA alleviates the side effect of hormone therapy while maintaining its therapeutic efficacy, which has great potential for developing the next generation of methods for osteoporosis treatment.
Keywords: osteoporosis, hormone therapy, upconversion nanoparticles, ethylenediaminetetraacetic acid
 Global reach, higher impact
Global reach, higher impact