13.3
Impact Factor
Theranostics 2020; 10(8):3451-3473. doi:10.7150/thno.39241 This issue Cite
Research Paper
A hypermethylation strategy utilized by enhancer-bound CARM1 to promote estrogen receptor α-dependent transcriptional activation and breast carcinogenesis
1. Fujian Provincial Key Laboratory of Innovative Drug Target Research, School of Pharmaceutical Sciences, Xiamen University, Xiang'an South Road, Xiamen, Fujian 361102, China
2. State Key Laboratory of Cellular Stress Biology, School of Pharmaceutical Sciences, Xiamen University, Xiang'an South Road, Xiamen, Fujian 361102, China
* These authors contributed equally
Abstract
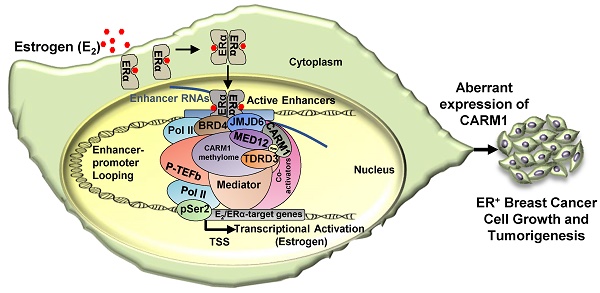
While protein arginine methyltransferases (PRMTs) and PRMT-catalyzed protein methylation have been well-known to be involved in a myriad of biological processes, their functions and the underlying molecular mechanisms in cancers, particularly in estrogen receptor alpha (ERα)-positive breast cancers, remain incompletely understood. Here we focused on investigating PRMT4 (also called coactivator associated arginine methyltransferase 1, CARM1) in ERα-positive breast cancers due to its high expression and the associated poor prognosis.
Methods: ChIP-seq and RNA-seq were employed to identify the chromatin-binding landscape and transcriptional targets of CARM1, respectively, in the presence of estrogen in ERα-positive MCF7 breast cancer cells. High-resolution mass spectrometry analysis of enriched peptides from anti-monomethyl- and anti-asymmetric dimethyl-arginine antibodies in SILAC labeled wild-type and CARM1 knockout cells were performed to globally map CARM1 methylation substrates. Cell viability was measured by MTS and colony formation assay, and cell cycle was measured by FACS analysis. Cell migration and invasion capacities were examined by wound-healing and trans-well assay, respectively. Xenograft assay was used to analyze tumor growth in vivo.
Results: CARM1 was found to be predominantly and specifically recruited to ERα-bound active enhancers and essential for the transcriptional activation of cognate estrogen-induced genes in response to estrogen treatment. Global mapping of CARM1 substrates revealed that CARM1 methylated a large cohort of proteins with diverse biological functions, including regulation of intracellular estrogen receptor-mediated signaling, chromatin organization and chromatin remodeling. A large number of CARM1 substrates were found to be exclusively hypermethylated by CARM1 on a cluster of arginine residues. Exemplified by MED12, hypermethylation of these proteins by CARM1 served as a molecular beacon for recruiting coactivator protein, tudor-domain-containing protein 3 (TDRD3), to CARM1-bound active enhancers to activate estrogen/ERα-target genes. In consistent with its critical role in estrogen/ERα-induced gene transcriptional activation, CARM1 was found to promote cell proliferation of ERα-positive breast cancer cells in vitro and tumor growth in mice.
Conclusions: our study uncovered a “hypermethylation” strategy utilized by enhancer-bound CARM1 in gene transcriptional regulation, and suggested that CARM1 can server as a therapeutic target for breast cancer treatment.
Keywords: Protein arginine methyltransferase, protein arginine methylation, tudor domain-containing protein, estrogen receptor, breast cancer
 Global reach, higher impact
Global reach, higher impact