13.3
Impact Factor
Theranostics 2020; 10(8):3533-3545. doi:10.7150/thno.39507 This issue Cite
Research Paper
Minimally invasive implantation and decreased inflammation reduce osteoinduction of biomaterial
1. State Key Laboratory Breeding Base of Basic Science of Stomatology (Hubei-MOST) and Key Laboratory of Oral Biomedicine, Ministry of Education, School and Hospital of Stomatology, Wuhan University, Wuhan 430079, China.
2. Department of Dental Implantology, School and Hospital of Stomatology, Wuhan University, Wuhan, 430079, China.
3. Institute of immunology, Shandong First Medical University & Shandong Academy of Medical Sciences, Taian 271000, China.
4. National Engineering Research Center for Nanomedicine, Hubei Key Laboratory of Bioinorganic Chemistry and Materia Medica, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan 430074, China.
5. Medical Research Institute, School of Medicine, Wuhan University, Wuhan 430071, China.
*These authors contributed equally to this work.
Abstract
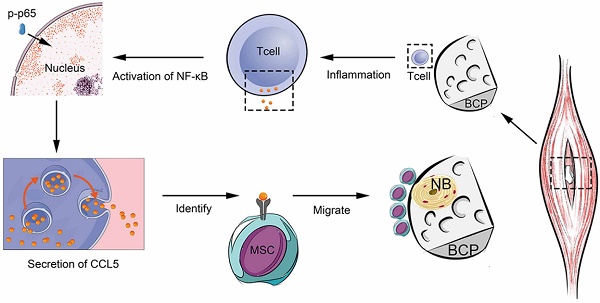
Surgical trauma of biomaterial implantation significantly influences the immune system and the biological effects of biomaterials. Minimally invasive surgery has become a trend of clinical development but violating the concept of osteoimmunomodulation will hinder the biological effects of materials. Our study focused on biphasic calcium phosphate (BCP), the ectopia osteoinductive materials, filling the research blank of the significance of adaptive immunity crosstalk with bone biomaterials, and improving the interaction mechanism between bone biomaterials and immune response.
Methods: The BCP bioceramics were implanted by conventional and minimally invasive methods in the gastrocnemius wild-type or T cells depleted mice to test the effect of ectopia osteoinduction. Moreover, flow cytometry was used to detect immune responses, T cell sorting and Western Blot molecular biology experiments, and transwell assays migration of mesenchymal stem cells (MSCs).
Results: We found that BCP, an implantable osteoinductive material, could not activate the adaptive immune response mediated by T cells after minimally invasive surgery. Further studies revealed that under the conventional non-minimally invasive BCP implantation, a positive correlation existed between T cell recruitment and the infiltration and osteogenic differentiation of MSCs. Interestingly, after BCP was implanted by minimally invasive surgery or implanted in T cell depleted mice, MSCs infiltration and osteogenic differentiation were significantly reduced, and BCP could not achieve the biological effects of ectopia ossification. Finally, we confirmed that a certain extent inflammatory stimulation activated the adaptive immune response mediated by T cells, up-regulated the nuclear factor-κB (NF-κB) signal in T cells, released a large amount of chemokine C-C motif chemokine ligand 5(CCL5) to recruit MSCs to the surrounding material, and finally achieved the ideal effect of osteoinduction.
Conclusion: From experimental research and clinical surgery, this study discovered that the T cells are indispensable in the ectopia ossification mediated by osteoinductive materials, put forward and confirmed the surgery method as a key variable factor restricting the application effect of biological materials, enriched the key mechanism of adaptive immunity in osteoimmunomodulation, and laid a theoretical foundation for the development of osteoinductive materials and bone tissue regeneration.
Keywords: osteoinduction, biomaterials, T cells, MSCs, CCL5, NF-κB pathway
 Global reach, higher impact
Global reach, higher impact