13.3
Impact Factor
Theranostics 2020; 10(8):3546-3561. doi:10.7150/thno.39786 This issue Cite
Research Paper
Targeted delivery of engineered auditory sensing protein for ultrasound neuromodulation in the brain
1. Department of Biomedical Engineering and Environmental Sciences, National Tsing Hua University, Hsinchu, Taiwan
2. Institute of Nuclear Engineering and Sciences, National Tsing Hua University, Hsinchu, Taiwan
3. Institute of Molecular Medicine, National Tsing Hua University, Hsinchu, Taiwan
4. Department of Molecular Medicine, National Tsing Hua University, Hsinchu, Taiwan
*C.-Y. Wu and C.-H. Fan contributed equally to this work.
Abstract
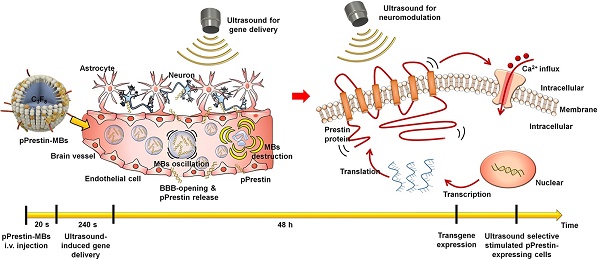
Sonogenetics is a promising approach for in vivo neuromodulation using ultrasound (US) to non-invasively stimulate cells in deep tissue. However, sonogenetics requires accurate transduction of US-responsive proteins into target cells. Here, we introduce a non-invasive and non-viral approach for intracerebral gene delivery. This approach utilizes temporary ultrasonic disruption of the blood-brain barrier (BBB) to transfect neurons at specific sites in the brain via DNA that encodes engineered US-responsive protein (murine Prestin (N7T, N308S))-loaded microbubbles (pPrestin-MBs). Prestin is a transmembrane protein that exists in the mammalian auditory system and functions as an electromechanical transducer. We further improved the US sensitivity of Prestin by introducing specific amino acid substitutions that frequently occur in sonar species into the mouse Prestin protein. We demonstrated this concept in mice using US with pPrestin-MBs to non-invasively modify and activate neurons within the brain for spatiotemporal neuromodulation.
Method: MBs composed of cationic phospholipid and C3F8 loaded with mouse Prestin plasmid (pPrestin) via electrostatic interactions. The mean concentration and size of the pPrestin-MBs were (16.0 ± 0.2) × 109 MBs/mL and 1.1 ± 0.2 μm, respectively. SH-SY5Y neuron-like cells and C57BL mice were used in this study. We evaluated the gene transfection efficiency and BBB-opening region resulting from pPrestin-MBs with 1-MHz US (pressure = 0.1-0.5 MPa, cycle = 50-10000, pulse repetition frequency (PRF): 0.5-5 Hz, sonication time = 60 s) using green fluorescence protein (Venus) and Evans blue staining.
Results: The maximum pPrestin expression with the highest cell viability occurred at a pressure of 0.5 MPa, cycle number of 5000, and PRF of 1 Hz. The cellular transfection rate with pPrestin-MBs and US was 20.2 ± 2.5%, which was 1.5-fold higher than that of commercial transfection agents (LT-1). In vivo data suggested that the most profound expression of pPrestin occurred at 2 days after performing pPrestin-MBs with US (0.5 MPa, 240 s sonication time). In addition, no server erythrocyte extravasations and apoptosis cells were observed at US-sonicated region. We further found that with 0.5-MHz US stimulation, cells with Prestin expression were 6-fold more likely to exhibit c-Fos staining than cells without Prestin expression.
Conclusion: Successful activation of Prestin-expressing neurons suggests that this technology provides non-invasive and spatially precise selective modulation of one or multiple specific brain regions.
Keywords: sonogenetic, neuromodulation, ultrasound, Prestin, microbubbles
 Global reach, higher impact
Global reach, higher impact