Impact Factor
Theranostics 2020; 10(8):3684-3707. doi:10.7150/thno.41580 This issue Cite
Review
Progress, opportunity, and perspective on exosome isolation - efforts for efficient exosome-based theranostics
1. Department of Neurosurgery of Hebi People's Hospital; Hebi Neuroanatomical Laboratory, Hebi, 458030, China.
2. School of Nursing, Zhengzhou University, Zhengzhou, 450001, China.
3. Henan Key Laboratory of Ion-beam Bioengineering, Zhengzhou University, Zhengzhou, China, 450000.
4. Guangdong Key Laboratory for Research and Development of Nature Drugs, School of Pharmacy, Guangdong Medical University, Zhanjiang 524023, China.
5. School of Statistics, Henan University of Economics and Law, Zhengzhou 450046, China.
6. Centre for Molecular Medicine and Innovative Therapeutics, Murdoch University, Perth 6150, Australia.
7. General Practice Centre, Nanhai Hospital, Southern Medical University, 528244, Foshan, China.
8. School of Medicine, Wake Forest University, Winston Salem, NC 27101, USA.
9. School of Medicine, and Centre for Molecular and Medical Research, Deakin University, 3216, Australia.
10. The First Affiliated Hospital of Zheng Zhou University, Zhengzhou 450001, China.
11. Perron Institute for Neurological and Translational Science, Perth 6009, Australia
#These authors contribute equally to this work.
Abstract
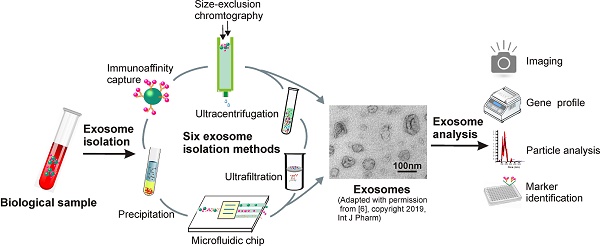
Exosomes are small extracellular vesicles with diameters of 30-150 nm. In both physiological and pathological conditions, nearly all types of cells can release exosomes, which play important roles in cell communication and epigenetic regulation by transporting crucial protein and genetic materials such as miRNA, mRNA, and DNA. Consequently, exosome-based disease diagnosis and therapeutic methods have been intensively investigated. However, as in any natural science field, the in-depth investigation of exosomes relies heavily on technological advances. Historically, the two main technical hindrances that have restricted the basic and applied researches of exosomes include, first, how to simplify the extraction and improve the yield of exosomes and, second, how to effectively distinguish exosomes from other extracellular vesicles, especially functional microvesicles. Over the past few decades, although a standardized exosome isolation method has still not become available, a number of techniques have been established through exploration of the biochemical and physicochemical features of exosomes. In this work, by comprehensively analyzing the progresses in exosome separation strategies, we provide a panoramic view of current exosome isolation techniques, providing perspectives toward the development of novel approaches for high-efficient exosome isolation from various types of biological matrices. In addition, from the perspective of exosome-based diagnosis and therapeutics, we emphasize the issue of quantitative exosome and microvesicle separation.
Keywords: Exosome, microvesicle, extracellular vesicle, microfluidic, diagnosis, separation
 Global reach, higher impact
Global reach, higher impact