Impact Factor
Theranostics 2020; 10(8):3793-3815. doi:10.7150/thno.40805 This issue Cite
Review
Comprehensive understanding of magnetic hyperthermia for improving antitumor therapeutic efficacy
1. Key Laboratory of Resource Biology and Biotechnology in Western China, Ministry of Education; School of Medicine, Northwest University, Xi'an 710069, China
2. CAS Key Laboratory for Biomedical Effects of Nanomaterials and Nanosafety, CAS Center for Excellence in Nanoscience, National Center for Nanoscience and Technology of China, No. 11, First North Road, Zhongguancun, Beijing 100190, China; University of Chinese Academy of Sciences, Beijing 100049, China
3. Key Laboratory of Synthetic and Natural Functional Molecule Chemistry of the Ministry of Education, College of Chemistry and Materials Science, Northwest University, Xi'an 710127, China
4. Beijing General Pharmaceutical Corporation, Beijing 100101, China
5. The National Institutes of Pharmaceutical R&D Co., Ltd., China Resources Pharmaceutical Group Limited, Beijing 102206, China
6. Beijing Institute of Traumatology and Orthopaedics, Beijing 100035, China
7. Neurophysiology Department, Beijing ChaoYang Emergency Medical Center, Beijing 100122, China
8. National Center for International Research of Biotargeting Theranostics, Guangxi Key Laboratory of Biotargeting Theranostics, Collaborative Innovation Center for Targeting Tumour Theranostics and Therapy, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
*These authors contributed equally to this work.
Abstract
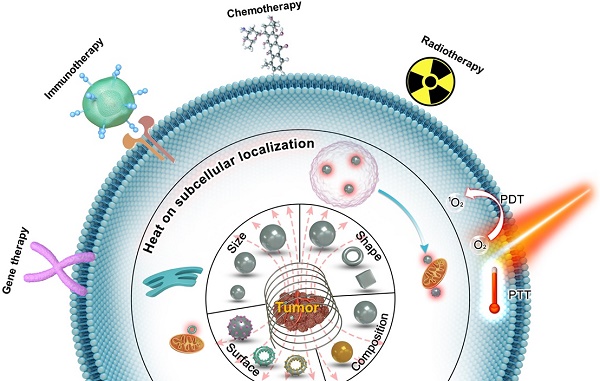
Magnetic hyperthermia (MH) has been introduced clinically as an alternative approach for the focal treatment of tumors. MH utilizes the heat generated by the magnetic nanoparticles (MNPs) when subjected to an alternating magnetic field (AMF). It has become an important topic in the nanomedical field due to their multitudes of advantages towards effective antitumor therapy such as high biosafety, deep tissue penetration, and targeted selective tumor killing. However, in order for MH to progress and to realize its paramount potential as an alternative choice for cancer treatment, tremendous challenges have to be overcome. Thus, the efficiency of MH therapy needs enhancement. In its recent 60-year of history, the field of MH has focused primarily on heating using MNPs for therapeutic applications. Increasing the thermal conversion efficiency of MNPs is the fundamental strategy for improving therapeutic efficacy. Recently, emerging experimental evidence indicates that MNPs-MH produces nano-scale heat effects without macroscopic temperature rise. A deep understanding of the effect of this localized induction heat for the destruction of subcellular/cellular structures further supports the efficacy of MH in improving therapeutic therapy. In this review, the currently available strategies for improving the antitumor therapeutic efficacy of MNPs-MH will be discussed. Firstly, the recent advancements in engineering MNP size, composition, shape, and surface to significantly improve their energy dissipation rates will be explored. Secondly, the latest studies depicting the effect of local induction heat for selectively disrupting cells/intracellular structures will be examined. Thirdly, strategies to enhance the therapeutics by combining MH therapy with chemotherapy, radiotherapy, immunotherapy, photothermal/photodynamic therapy (PDT), and gene therapy will be reviewed. Lastly, the prospect and significant challenges in MH-based antitumor therapy will be discussed. This review is to provide a comprehensive understanding of MH for improving antitumor therapeutic efficacy, which would be of utmost benefit towards guiding the users and for the future development of MNPs-MH towards successful application in medicine.
Keywords: magnetic nanoparticles, magnetic hyperthermia, macroscopic heating, local induction heat, synergistic strategy
 Global reach, higher impact
Global reach, higher impact