13.3
Impact Factor
Theranostics 2020; 10(10):4530-4543. doi:10.7150/thno.39700 This issue Cite
Research Paper
A dual-functional Embolization-Visualization System for Fluorescence image-guided Tumor Resection
1. Department of Bioengineering, University of Utah, Salt Lake City, UT, 84112 USA.
2. Utah Center for Nanomedicine, Nano Institute of Utah, University of Utah, Salt Lake City, UT, 84112 USA.
3. Department of Pharmaceutics and Pharmaceutical Chemistry, University of Utah, Salt Lake City, UT, 84112 USA.
4. Department of Surgery, Division of Otolaryngology-Head and Neck Surgery, University of Utah School of Medicine, Salt Lake City, UT 84113.
Abstract
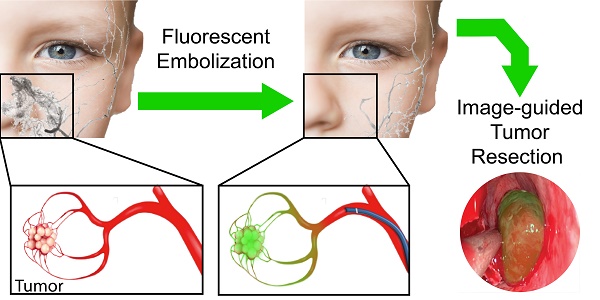
Rationale: Intraoperative bleeding impairs physicians' ability to visualize the surgical field, leading to increased risk of surgical complications and reduced outcomes. Bleeding is particularly challenging during endoscopic-assisted surgical resection of hypervascular tumors in the head and neck. A tool that controls bleeding while marking tumor margins has the potential to improve gross tumor resection, reduce surgical morbidity, decrease blood loss, shorten procedure time, prevent damage to surrounding tissues, and limit postoperative pain. Herein, we develop and characterize a new system that combines pre-surgical embolization with improved visualization for endoscopic fluorescence image-guided tumor resection.
Methods: Silk-elastinlike protein (SELP) polymers were employed as liquid embolic vehicles for delivery of a clinically used near-infrared dye, indocyanine green (ICG). The biophysical properties of SELP, including gelation kinetics, modulus of elasticity, and viscosity, in response to ICG incorporation using rheology, were characterized. ICG release from embolic SELP was modeled in tissue phantoms and via fluorescence imaging. The embolic capability of the SELP-ICG system was then tested in a microfluidic model of tumor vasculature. Lastly, the cytotoxicity of the SELP-ICG system in L-929 fibroblasts and human umbilical vein endothelial cells (HUVEC) was assessed.
Results: ICG incorporation into SELP accelerated gelation and increased its modulus of elasticity. The SELP embolic system released 83 ± 8% of the total ICG within 24 hours, matching clinical practice for pre-surgical embolization procedures. Adding ICG to SELP did not reduce injectability, but did improve the gelation kinetics. After simulated embolization, ICG released from SELP in tissue phantoms diffused a sufficient distance to deliver dye throughout a tumor. ICG-loaded SELP was injectable through a clinical 2.3 Fr microcatheter and demonstrated deep penetration into 50-µm microfluidic-simulated blood vessels with durable occlusion. Incorporation of ICG into SELP improved biocompatibility with HUVECs, but had no effect on L-929 cell viability.
Principle Conclusions: We report the development and characterization of a new, dual-functional embolization-visualization system for improving fluorescence-imaged endoscopic surgical resection of hypervascular tumors.
Keywords: Embolic, Image-Guided Surgery, Indocyanine Green, Silk-Elastinlike Protein Polymer
 Global reach, higher impact
Global reach, higher impact