13.3
Impact Factor
Theranostics 2020; 10(14):6136-6148. doi:10.7150/thno.45973 This issue Cite
Research Paper
Fluorescence-guided fiber-optic micronavigation using microscopic identification of vascular boundary of liver segment and tumors
1. Department of General, Visceral and Transplantation Surgery, University Hospital Heidelberg, Heidelberg, Germany
2. Department of General Surgery, The Third Affiliated Hospital of Sun Yat-sen University, Guangzhou, China
Abstract
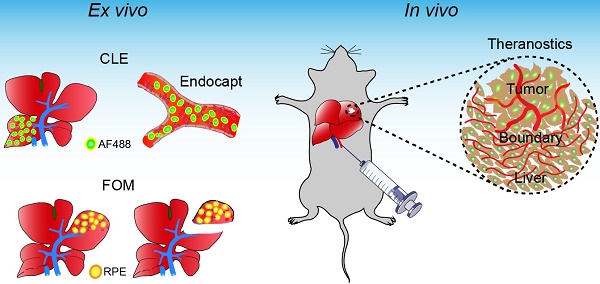
Background: The exact identification of tumor boundaries and related liver segments is especially important for liver tumor surgery. This study aimed to evaluate a new approach for vascular boundary assessment and surgical navigation based on fiber-optic microscopy and microvascular fluorescence labeling.
Methods: Antibody clones with fast binding ability were identified and selected using immunofluorescence. We evaluated the endothelial capture efficacy for an anti-mouse CD31 antibody labeled with different fluorophores and different degrees of labeling ex vivo. Segment boundary identification and navigation potential using endothelial capture were explored by two different fiber-optic microscopy systems. Finally, microvasculature labeling and fiber-optic microscopy were used to identify and treat microscopic liver tumors in vivo.
Results: The following monoclonal antibodies were selected: anti-mouse CD31 (clone 390), anti-mouse CD54 (YN1/1.7.4), anti-human CD31 (WM59), and anti-human CD54 (HA58). These clones showed fast binding to endothelial cells and had long half-lives. The fluorophore choice and the degree of antibody labeling did not significantly affect capture efficacy in an isolated liver perfusion model. The microvascular system was clearly identified with wide-field fiber-optic microscopy after labeling the endothelium with low doses of specific antibodies, and the specifically labeled liver segment could be microscopically dissected. High antibody doses were required for confocal laser endomicroscopy. After microscopically identifying the vascular margin in vivo, tumor thermoablation strongly reduced tumor size or totally eliminated tumors.
Conclusions: We demonstrated that vascular boundaries of liver tumors and locally perfused liver segments were accurately identified and surgical micronavigation was facilitated with fiber-optic microscopy and selected endothelium-specific antibodies.
Keywords: Vascular boundary, Fluorescence imaging, Endothelial capture, Surgical micronavigation, Fiber-optic microscopy
 Global reach, higher impact
Global reach, higher impact