13.3
Impact Factor
Theranostics 2020; 10(14):6149-6166. doi:10.7150/thno.46053 This issue Cite
Research Paper
DR-region of Na+/K+-ATPase is a target to ameliorate hepatic insulin resistance in obese diabetic mice
1. Department of Pharmacology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore.
2. Center of Clinical Research, Wuxi People's Hospital of Nanjing Medical University, Wuxi, Jiangsu 214023, PR China; Lung Transplant Group, Wuxi People's Hospital Affiliated to Nanjing Medical University, Wuxi, Jiangsu 214023, PR China.
3. National University of Singapore (Suzhou) Research Institute, Suzhou, China.
Abstract
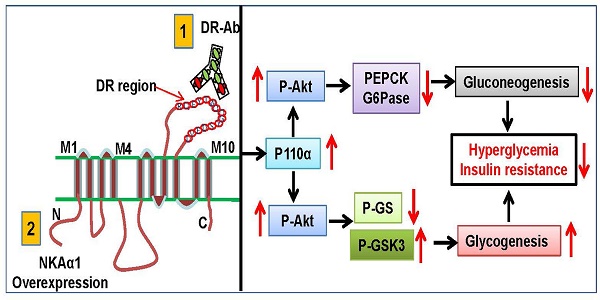
Reduced hepatic Na+/K+-ATPase (NKA) activity and NKAα1 expression are engaged in the pathologies of metabolism diseases. The present study was designed to investigate the potential roles of NKAα1 in hepatic gluconeogenesis and glycogenesis in both hepatocytes and obese diabetic mice.
Methods: Insulin resistance was mimicked by glucosamine (GlcN) in either human hepatocellular carcinoma (HepG2) cells or primary mouse primary hepatocytes. Obese diabetic mice were induced by high-fat diet (HFD) feeding for 12 weeks.
Results: We found that both NKA activity and NKAα1 protein level were downregulated in GlcN-treated hepatocytes and in the livers of obese diabetic mice. Pharmacological inhibition of NKA with ouabain worsened, while activation of NKAα1 with an antibody against an extracellular DR region of NKAα1 subunit (DR-Ab) prevented GlcN-induced increase in gluconeogenesis and decrease in glycogenesis. Likewise, the above results were also corroborated by the opposite effects of genetic knockout/overexpression of NKAα1 on both gluconeogenesis and glycogenesis. In obese diabetic mice, hepatic activation or overexpression of NKAα1 stimulated the PI3K/Akt pathway to suppress hyperglycemia and improve insulin resistance. More importantly, loss of NKA activities in NKAα1+/- mice was associated with more susceptibility to insulin resistance following HFD feeding.
Conclusions: Our findings suggest that NKAα1 is a physiological regulator of glucose homoeostasis and its DR-region is a novel target to treat hepatic insulin resistance.
Keywords: diabetes, insulin resistance, Na+/K+-ATPase, gluconeogenesis, glycogenesis
 Global reach, higher impact
Global reach, higher impact