13.3
Impact Factor
Theranostics 2020; 10(14):6182-6200. doi:10.7150/thno.46553 This issue Cite
Research Paper
Kindlin-2 deficiency induces fatal intestinal obstruction in mice
1. Department of Human Anatomy, Histology and Embryology, Key Laboratory of Carcinogenesis and Translational Research, Ministry of Education, and State Key Laboratory of Natural and Biomimetic Drugs, Peking University Health Science Center, Beijing 100191, China
2. Department of Physiology and Pathophysiology, School of Basic Medical Sciences, Peking University Health Science Center, Beijing 100191, China
3. Department of Surgery and Interdepartmental Program in Vascular Biology and Therapeutics, Yale University School of Medicine, New Haven, Connecticut, USA
Abstract
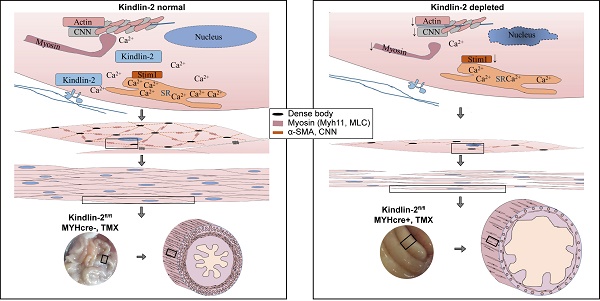
Rationale: Smooth muscle-motility disorders are mainly characterized by impaired contractility and functional intestinal obstruction. Some of these cases are caused by genetic mutations of smooth muscle genes ACTA2, ACTG2, MYH11, MYLK and LMOD1. Still the etiology is complex and multifactorial and the underlying pathology is poorly understood. Integrin interaction protein Kindlin-2 is widely expressed in striated and smooth muscle cells (SMC). However, the function of Kindlin-2 in the smooth muscle remains elusive.
Methods: We generated two mouse models using different cre promoter transgenic mice, Kindlin-2fl/fl SM22α-cre+ (cKO mice) and Kindlin-2fl/fl; MYH-cre+ (iKO mice). Embryos and adult tissues were prepared for hematoxylin and eosin (H&E) staining, immunohistochemistry (IHC) and terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) apoptosis assay. We investigated ultrastructure changes of mouse smooth muscle using transmission electron microscopy (TEM) and measured smooth muscle contractile force in mounting aortic and intestinal rings using the multiwire myograph system (DMT 620M). In addition, cell traction force microscopy (CTFM) was applied to observe the functional change of primary SMC after Kindlin-2 depletion by RNAi.
Results: Depletion of Kindlin-2 encoding gene Fermt2 in embryonic smooth muscles leads to apoptosis, downregulates the key components of SMC, impairs smooth muscle development, and finally causes embryonic death at E14.5. Tamoxifen-induced Kindlin-2-specific knockout in adult mouse smooth muscle showed decreased blood pressure, intestinal hypoperistalsis, and eventually died of intestinal obstruction. Kindlin-2 depletion also leads to downregulated Myh11, α-SMA, and CNN, shortened myofilament, broken myofibrils, and impaired contractility of the smooth muscles in iKO mice. Mechanistically, loss of Kindlin-2 decreases Ca2+ influx in primary vascular smooth muscle cells (PVSMC) by downregulating the expression of calcium-binding protein S100A14 and STIM1.
Conclusion: We demonstrated that Kindlin-2 is essential for maintaining the normal structure and function of smooth muscles. Loss of Kindlin-2 impairs smooth muscle formation during embryonic development by inducing apoptosis and jeopardizes the contraction of adult smooth muscle by blocking Ca2+ influx that leads to intestinal obstruction. Mice with Kindlin-2 depletion in adult smooth muscle could be a potent animal model of intestinal obstruction for disease research, drug treatment and prognosis.
Keywords: Kindlin-2, Smooth muscle structure, Smooth muscle contraction, Intestinal obstruction
 Global reach, higher impact
Global reach, higher impact