13.3
Impact Factor
Theranostics 2020; 10(14):6322-6336. doi:10.7150/thno.42581 This issue Cite
Research Paper
Faster, sharper, more precise: Automated Cluster-FLIM in preclinical testing directly identifies the intracellular fate of theranostics in live cells and tissue
1. Institute of Experimental Physics, Freie Universität Berlin, Arnimallee 14, 14195 Berlin, Germany
2. Institute of Pharmacy (Pharmacology and Toxicology), Freie Universität Berlin, Königin-Luise-Str. 2+4, 14195 Berlin, Germany
3. Institute of Chemistry and Biochemistry, Freie Universität Berlin, Takustr. 3, 14195 Berlin, Germany
* contributed equally
Abstract
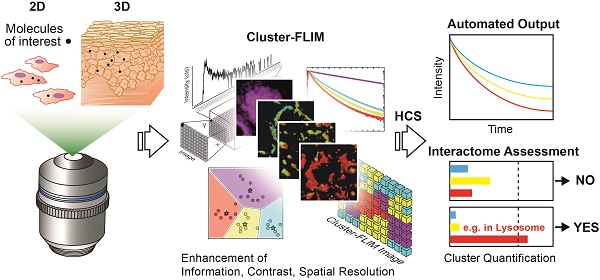
Fluorescence microscopy is widely used for high content screening in 2D cell cultures and 3D models. In particular, 3D tissue models are gaining major relevance in modern drug development. Enabling direct multiparametric evaluation of complex samples, fluorescence lifetime imaging (FLIM) adds a further level to intensity imaging by the sensitivity of the fluorescence lifetime to the microenvironment. However, the use of FLIM is limited amongst others by the acquisition of sufficient photon numbers without phototoxic effects in live cells. Herein, we developed a new cluster-based analysis method to enhance insight, and significantly speed up analysis and measurement time for the accurate translation of fluorescence lifetime information into pharmacological pathways.
Methods: We applied a fluorescently-labeled dendritic core-multishell nanocarrier and its cargo Bodipy as molecules of interest (MOI) to human cells and reconstructed human tissue. Following the sensitivity and specificity assessment of the fitting-free Cluster-FLIM analysis of data in silico and in vitro, we evaluated the dynamics of cellular molecule uptake and intracellular interactions. For 3D live tissue investigations, we applied multiphoton (mp) FLIM. Owing to Cluster-FLIM's statistics-based fitting-free analysis, we utilized this approach for automatization.
Results: To discriminate the fluorescence lifetime signatures of 5 different fluorescence species in a single color channel, the Cluster-FLIM method requires only 170, respectively, 90 counts per pixel to obtain 95% sensitivity (hit rate) and 95% specificity (correct rejection rate). Cluster-FLIM revealed cellular interactions of MOIs, representing their spatiotemporal intracellular fate. In a setting of an automated workflow, the assessment of lysosomal trapping of the MOI revealed relevant differences between normal and tumor cells, as well as between 2D and 3D models.
Conclusion: The automated Cluster-FLIM tool is fitting-free, providing images with enhanced information, contrast, and spatial resolution at short exposure times and low fluorophore concentrations. Thereby, Cluster-FLIM increases the applicability of FLIM in high content analysis of target molecules in drug development and beyond.
Keywords: fluorescence lifetime imaging microscopy, high throughput imaging, FLIM automatization, nanomedicine, drug development
 Global reach, higher impact
Global reach, higher impact