Impact Factor
Theranostics 2020; 10(15):6615-6628. doi:10.7150/thno.43169 This issue Cite
Research Paper
Mesenchymal stem cells injected into carotid artery to target focal brain injury home to perivascular space
1. NeuroRepair Department, Mossakowski Medical Research Centre, PAS, Warsaw, Poland
2. Department of Neurosurgery, Central Clinical Hospital of Ministry of the Interior and Administration, Warsaw, Poland
3. Center for Advanced Imaging Research, Department of Diagnostic Radiology and Nuclear Medicine, University of Maryland Marlene and Stewart Greenebaum Comprehensive Cancer Center, University of Maryland, Baltimore, MD, USA
4. Tumor Immunology and Immunotherapy Program, University of Maryland Marlene and Stewart Greenebaum Comprehensive Cancer Center, University of Maryland, Baltimore, MD, USA
Abstract
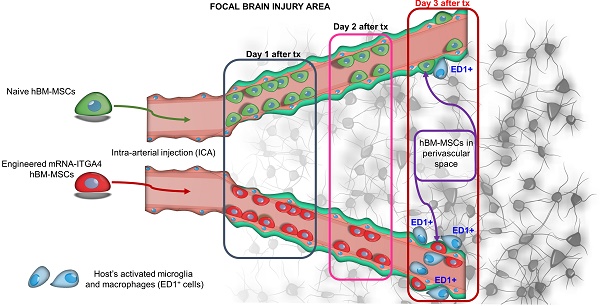
Rationale: The groundbreaking discovery of mesenchymal stem cells (MSCs) with their multifaceted benefits led to their widespread application in experimental medicine, including neurology. Efficient delivery of MSCs to damaged regions of the central nervous system may be a critical factor in determining outcome. Integrin VLA-4 (α4β1) coded by ITGA4 and ITGB1 genes is an adhesion molecule expressed by leukocytes, which is responsible for initiation of their diapedesis through cell docking to the inflamed vessel wall expressing VCAM1 receptor. This function of VLA-4 has been recapitulated in neural stem cells and glial progenitors. Thus, it was prudent to investigate this tool as a vehicle driving extravasation of MSCs. Since MSCs naturally express ITGB1 subunit, we decided to supplement them with ITGA4 only. The purpose of our current study is to investigate the eventual fate of IA delivered ITGA4 engineered and naive MSCs.
Methods: mRNA-ITGA4 transfected and naive MSCs were injected to right internal carotid artery of rats with focal brain injury. Through next three days MSC presence in animals' brain was navigated by magnetic resonance imaging. Transplanted cell location relative to the brain blood vessels and host immunological reaction were analyzed post-mortem by immunohistochemistry. The chemotaxis of modified and naive MSCs was additionally examined in in vitro transwell migration assay.
Results: Both naïve and ITGA4-overexpressing cells remained inside the vascular lumen over the first two days after IA infusion. On the third day, 39% of mRNA-ITGA4 modified and 51% naïve MSCs homed to perivascular space in the injury region (p=NS). The gradual decrease of both naive and mRNA-ITGA4 transfected hBM-MSCs in the rat brain was observed. mRNA-ITGA4 transfected MSCs appeared to be more vulnerable to phagocytosis than naïve cells. Moreover, in vitro study revealed that homogenate from the injured brain repels migration of MSCs, corroborating the incomplete extravasation observed in vivo.
Conclusions: In summary, IA transplanted MSCs are capable of homing to the perivascular space, an integral part of neurovascular unit, which might contribute to the replacement of injured pericytes, a critical element facilitating restoration of CNS function. The mRNA-ITGA4 transfection improves cell docking to vessel but this net benefit vanishes over the next two days due to fast clearance from cerebral vessels of the majority of transplanted cells, regardless of their engineering status. The drawbacks of mRNA-ITGA4 transfection become apparent on day 3 post transplantation due to the lower survival and higher vulnerability to host immune attack.
Keywords: mesenchymal stem cells, integrin VLA4 (α4β1), mRNA cell transfection, brain injury, intra-arterial delivery, perivascular space
 Global reach, higher impact
Global reach, higher impact