13.3
Impact Factor
Theranostics 2020; 10(17):7492-7509. doi:10.7150/thno.44309 This issue Cite
Research Paper
MicroRNA-181 regulates the development of Ossification of Posterior longitudinal ligament via Epigenetic Modulation by targeting PBX1
1. Department of Orthopedics, Changzheng Hospital Affiliated to Second Military Medical University, 415th Feng Yang Road, Shanghai, 200003, PR China.
2. Undergraduate Brigade, Changhai Hospital Affiliated to Second Military Medical University, 168th Chang Hai Road, Shanghai, 200433, China.
3. Research Center of Developmental Biology, Second Military Medical University, 800th Xiang Yin Road, Shanghai, 200433, PR China.
*These authors contributed equally to this work.
Abstract
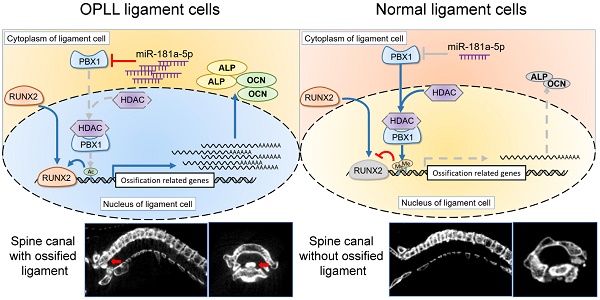
Objectives: Ossification of the posterior longitudinal ligament (OPLL) presents as the development of heterotopic ossification in the posterior longitudinal ligament of the spine. The etiology of OPLL is genetically linked, as shown by its high prevalence in Asian populations. However, the molecular mechanism of the disease remains obscure. In this study, we explored the function and mechanism of OPLL-specific microRNAs.
Methods: The expression levels of the ossification-related OPLL-specific miR-181 family were measured in normal or OPLL ligament tissues. The effect of miR-181a on the ossification of normal or pathogenic ligament cells was tested using real-time polymerase chain reaction (PCR), Western blot, alizarin red staining and alkaline phosphatase (ALP) staining. The candidate targets of miR-181 were screened using a dual luciferase reporter assay and functional analysis. The link between miR-181a and its target PBX1 was investigated using chromatin immunoprecipitation, followed by real-time PCR detection. Histological and immunohistochemical analysis as well as micro-CT scanning were used to evaluate the effects of miR-181 and its antagonist using both tip-toe-walking OPLL mice and in vivo bone formation assays.
Results: Using bioinformatic analysis, we found that miR-181a-5p is predicted to play important roles in the development of OPLL. Overexpression of miR-181a-5p significantly increased the expression of ossification-related genes, staining level of alizarin red and ALP activity, while the inhibition of miR-181a-5p by treatment with an antagomir had the opposite effects. Functional analysis identified PBX1 as a direct target of miR-181a-5p, and we determined that PBX1 was responsible for miR-181a-5p's osteogenic phenotype. By chromatin immunoprecipitation assay, we found that miR-181a-5p controls ligament cell ossification by regulating PBX1-mediated modulation of histone methylation and acetylation levels in the promoter region of osteogenesis-related genes. Additionally, using an in vivo model, we confirmed that miR-181a-5p can substantially increase the bone formation ability of posterior ligament cells and cause increased osteophyte formation in the cervical spine of tip-toe-walking mice.
Conclusions: Our data unveiled the mechanism by which the miR-181a-5p/PBX1 axis functions in the development of OPLL, and it revealed the therapeutic effects of the miR-181a-5p antagomir in preventing OPLL development both in vivo and in vitro. Our work is the first to demonstrate that microRNA perturbation could modulate the development of OPLL through epigenetic regulation.
Keywords: OPLL, miR-181, PBX1, ossification, histone modification
 Global reach, higher impact
Global reach, higher impact