13.3
Impact Factor
Theranostics 2020; 10(17):7527-7544. doi:10.7150/thno.45989 This issue Cite
Research Paper
LncRNA CSMD1-1 promotes the progression of Hepatocellular Carcinoma by activating MYC signaling
1. State Key Laboratory of Oncology in South China, and Collaborative Innovation Center for Cancer Medicine, Sun Yat-Sen University Cancer Center, Guangzhou, China 510060.
2. Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, China 510095.
3. Central Laboratory for Medical Research, Shanghai Tenth People's Hospital, Tongji University School of Medicine.
4. Department of Hepatobiliary Surgery, Sun Yat-Sen University Cancer Center, Guangzhou, China 510060.
5. Department of Pathology, Sun Yat-Sen University Cancer Center, Guangzhou, China 510060.
6. Department of Oncology, The Second Affiliated Hospital of Nanchang University, Nanchang, China.
*These authors contributed equally to this work.
Abstract
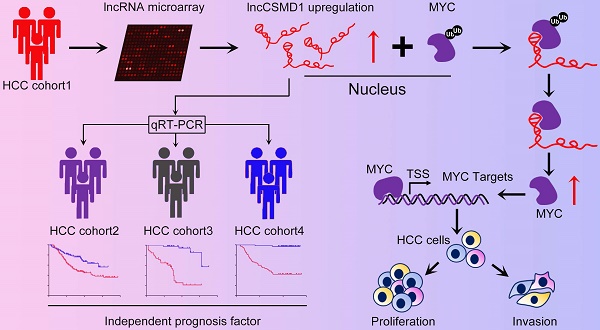
Emerging evidence suggests that long non-coding RNAs (lncRNA) play critical roles in the development and progression of diverse cancers including hepatocellular carcinoma (HCC), but the underlying molecular mechanisms of lncRNAs that are involved in hepatocarcinogenesis have not been fully explored.
Methods: In this study, we profiled lncRNA expression in 127 pairs of HCC and nontumor liver tissues (a Discovery Cohort) using a custom microarray. The expression and clinical significance of lncCSMD1-1 were then validated with qRT-PCR and COX regression analysis in a Validation Cohort (n=260) and two External Validation Cohorts (n=92 and n=124, respectively). In vitro and in vivo assays were performed to explore the biological effects of lncCSMD1-1 on HCC cells. The interaction of lncCSMD1-1 with MYC was identified by RNA pull-down and RNA immunoprecipitation. The role of LncCSMD1-1 in the degradation of MYC protein was also investigated.
Results: With microarray, we identified a highly upregulated lncRNA, lncCSMD1-1, which was associated with tumor progression and poor prognosis in the Discovery Cohort, and validated in another 3 HCC cohorts. Consistently, ectopic expression of lncCSMD1-1 notably promotes cell proliferation, migration, invasion, tumor growth and metastasis of HCC cells in in vitro and in vivo experiments. Gene expression profiling on HCC cells and gene sets enrichment analysis indicated that the MYC target gene set was significantly enriched in HCC cells overexpressing lncCSMD1-1, and lncCSMD1-1 was found to directly bind to MYC protein in the nucleus of HCC cells, which resulted in the elevation of MYC protein. Mechanistically, lncCSMD1-1 interacted with MYC protein to block its ubiquitin-proteasome degradation pathway, leading to activation of its downstream target genes.
Conclusion: lncCSMD1-1 is upregulated in HCC and promotes progression of HCC by activating the MYC signaling pathway. These results provide the evidence that lncCSMD1-1 may serve as a novel prognostic marker and potential therapeutic target for HCC.
Keywords: Long noncoding RNA, lncCSMD1-1, Hepatocellular carcinoma, MYC, prognosis
 Global reach, higher impact
Global reach, higher impact