Impact Factor
Theranostics 2020; 10(17):7645-7655. doi:10.7150/thno.44556 This issue Cite
Research Paper
Molecular analysis of circulating tumor cells of metastatic castration-resistant Prostate Cancer Patients receiving 177Lu-PSMA-617 Radioligand Therapy
1. Department of Nuclear Medicine, University Hospital Münster, Münster, Germany.
2. Department of Nuclear Medicine, University Hospital Essen, Essen, Germany.
3. Department of Urology, University Hospital Münster, Münster, Germany.
4. West German Cancer Center, Münster.
*equal contribution
Abstract
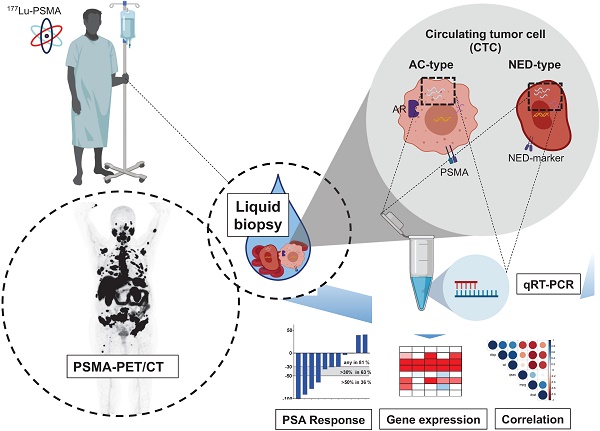
Rationale: Lu-177-PSMA-617 radioligand therapy (RLT) is currently under approval for treatment of metastatic castration resistant prostate cancer (mCRPC) patients with late stage disease. However, previous studies demonstrated both heterogeneity of prostate specific membrane antigen (PSMA) expression, as well as response to PSMA treatment among mCRPC patients. Thus, there is an unmet need for identifying predictive parametres prior or under PSMA-RLT treatment. We therefore aimed to correlate several clinical and molecular parameters with response to PSMA treatment in a cohort of mCRPC patients undergoing PSMA RLT followed by a detailed analysis of promising candidates.
Methods: Nineteen patients, median age 68.8 years (range: 56.9 - 83.3) with mCRPC were included in this study. We performed baseline analysis of clinical parameters based on PSMA PET/CT, (metabolic tumor volume (MTV), total tumor volume (TTV)), serum PSA, ALP, LDH and gene expression analysis of circulating tumor cells (expression of AR full length (AR-FL), AR splice variant 7 (AR-V7), PSA and PSMA) as well as common markers for neuroendocrine differentiation (NED).
Results: Patients presented with bone, lymph node, and visceral metastases (89%, 68%, and 21%, respectively). All patients were pretreated with docetaxel, either abiraterone or enzalutamide, or both. Biochemical response in terms of PSA decline ≥50 or ≥30% was observed in 42% and 63%, respectively. There were significant correlations between PSA and PSMA mRNA expression, as well as tumor volumes (both MTV and TTV), AR-FL and AR-V7 mRNA expression. However, there was no correlation with response to PSMA treatment. Furthermore, none of these parameters was significantly correlated with baseline serum PSA values. Common NED markers were shown to be specifically high expressed and revealed impact on OS independent from AR-V7 gene expression.
Conclusion: We demonstrate that AR-FL and its splice variant AR-V7 might serve as prognostic biomarkers displaying high tumor burden in mCRPC patient prior to PSMA-RLT. Contrary, PSMA, which has been discussed as a biomarker for PSMA targeted treatment, does not display strong prognostic ability - at least on the mRNA level. Surprisingly, none of these parameters correlates to response to PSMA treatment. In contrast, commom NED markers such as SYP and ENO2 as well as FOXA1 expression level seem to predict OS, but not PFS, more reliably. We admit that a limitation of our study is the focus on mRNA expression of potential biomarkers only. Further investigations analyzing the potential role of protein expression of these markers are therefore warranted.
Keywords: CTC, PSMA, 177Lu-PSMA-617, prostate cancer
 Global reach, higher impact
Global reach, higher impact