13.3
Impact Factor
Theranostics 2020; 10(17):7857-7871. doi:10.7150/thno.43154 This issue Cite
Research Paper
Protective effect of Soluble Epoxide Hydrolase Inhibition in Retinal Vasculopathy associated with Polycystic Kidney Disease
1. 5th Medical Department, Medical Faculty Mannheim, University of Heidelberg, D-68167 Mannheim, Germany.
2. Institute for Vascular Signalling, Center for Molecular Medicine, Goethe University, D-60590 Frankfurt, Germany.
3. Sanofi Research and Development, Industriepark Hoechst, D-65926 Frankfurt, Germany.
4. Institute of Experimental and Clinical Pharmacology and Toxicology, Medical Faculty Mannheim, University of Heidelberg, D-68169 Mannheim, Germany.
5. Center of Medical Research, Medical Faculty Mannheim, University of Heidelberg, D-68167 Mannheim, Germany.
6. Center of Medical Research, Medical Faculty Mannheim, University of Heidelberg, D-68167 Mannheim, Germany.
*These authors contributed equally to this study.
Abstract
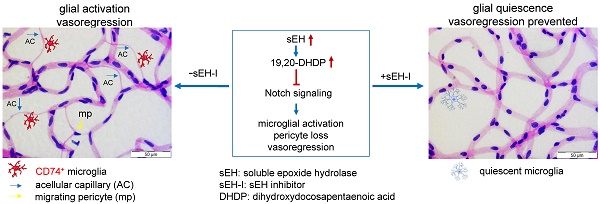
Rationale: Vasoregression secondary to glial activation develops in various retinal diseases, including retinal degeneration and diabetic retinopathy. Photoreceptor degeneration and subsequent retinal vasoregression, characterized by pericyte loss and acellular capillary formation in the absence diabetes, are also seen in transgenic rats expressing the polycystic kidney disease (PKD) gene. Activated Müller glia contributes to retinal vasodegeneration, at least in part via the expression of the soluble epoxide hydrolase (sEH). Given that an increase in sEH expression triggered vascular destabilization in diabetes, and that vasoregression is similar in diabetic mice and PKD rats, the aim of the present study was to determine whether sEH inhibition could prevent retinal vasoregression in the PKD rat.
Methods: One-month old male homozygous transgenic PKD rats were randomly allocated to receive vehicle or a sEH inhibitor (sEH-I; Sar5399, 30 mg/kg) for four weeks. Wild-type Sprague-Dawley (SD) littermates received vehicle as controls. Retinal sEH expression and activity were measured by Western blotting and LC-MS, and vasoregression was quantified in retinal digestion preparations. Microglial activation and immune response cytokines were assessed by immunofluorescence and quantitative PCR, respectively. 19,20-dihydroxydocosapentaenoic acid (19,20-DHDP) mediated Notch signaling, microglial activation and migration were assessed in vivo and in vitro.
Results: This study demonstrates that sEH expression and activity were increased in PKD retinae, which led to elevated production of 19,20-DHDP and the depression of Notch signaling. The latter changes elicited pericyte loss and the recruitment of CD11b+/CD74+ microglia to the perivascular region. Microglial activation increased the expression of immune-response cytokines, and reduced levels of Notch3 and delta-like ligand 4 (Dll4). Treatment with Sar5399 decreased 19,20-DHDP generation and increased Notch3 expression. Sar5399 also prevented vasoregression by reducing pericyte loss and suppressed microglial activation as well as the expression of immune-response cytokines. Mechanistically, the activation of Notch signaling by Dll4 maintained a quiescent microglial cell phenotype, i.e. reduced both the surface presentation of CD74 and microglial migration. In contrast, in retinal explants, 19,20-DHDP and Notch inhibition both promoted CD74 expression and reversed the Dll4-induced decrease in migration.
Conclusions: Our data indicate that 19,20-DHDP-induced alterations in Notch-signaling result in microglia activation and pericyte loss and contribute to retinal vasoregression in polycystic kidney disease. Moreover, sEH inhibition can ameliorate vasoregression through reduced activity of inflammatory microglia. sEH inhibition is thus an attractive new therapeutic approach to prevent retinal vasoregression.
Keywords: retinal vasoregression, sEH inhibition, CD74, 19, 20-DHDP, neurovascular unit
 Global reach, higher impact
Global reach, higher impact