Impact Factor
Theranostics 2020; 10(18):7974-7992. doi:10.7150/thno.45037 This issue Cite
Research Paper
RNA-binding protein NONO contributes to cancer cell growth and confers drug resistance as a theranostic target in TNBC
1. Department of Convergence Medicine, University of Ulsan College of Medicine, Seoul, Korea.
2. Asan Institute for Life Sciences, Asan Medical Center, Seoul, Korea.
3. Department of Immunology School of Medicine, Konkuk University, Chungju, South Korea.
4. QIMR Berghofer Medical Research Institute, Brisbane, QLD, Australia.
5. MTA TTK Lendület Cancer Biomarker Research Group; Institute of Enzymology; Semmelweis University 2nd Dept. of Pediatrics, Budapest, Hungary.
6. Department of Pathology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea.
7. Department of Cell, Development and Cancer Biology, Oregon Health & Science University, Portland, OR, USA.
8. Department of Life Science, Chung-Ang University, Seoul, Korea.
Abstract
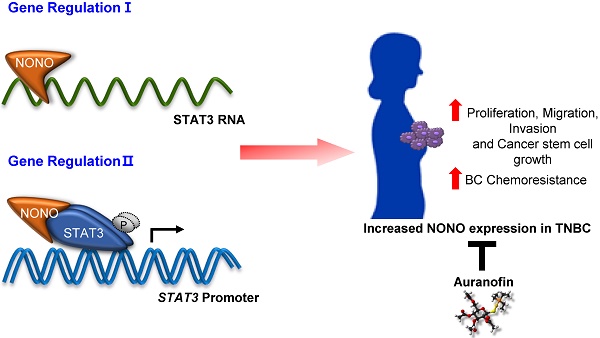
Breast cancer (BC) is one of the most common cancers in women. TNBC (Triple-negative breast cancer) has limited treatment options and still lacks viable molecular targets, leading to poor outcomes. Recently, RNA-binding proteins (RBPs) have been shown to play crucial roles in human cancers, including BC, by modulating a number of oncogenic phenotypes. This suggests that RBPs represent potential molecular targets for BC therapy.
Methods: We employed genomic data to identify RBPs specifically expressed in TNBC. NONO was silenced in TNBC cell lines to examine cell growth, colony formation, invasion, and migration. Gene expression profiles in NONO-silenced cells were generated and analyzed. A high-throughput screening for NONO-targeted drugs was performed using an FDA-approved library.
Results: We found that the NONO RBP is highly expressed in TNBC and is associated with poor patient outcomes. NONO binds to STAT3 mRNA, increasing STAT3 mRNA levels in TNBC. Surprisingly, NONO directly interacts with STAT3 protein increasing its stability and transcriptional activity, thus contributing to its oncogenic function. Importantly, high-throughput drug screening revealed that auranofin is a potential NONO inhibitor and inhibits cell growth in TNBC.
Conclusions: NONO is an RBP upstream regulator of both STAT3 RNA and protein levels and function. It represents an important and clinically relevant promoter of growth and resistance of TNBCs. NONO is also therefore a potential therapeutic target in TNBC.
Keywords: RBP, NONO, TNBC, STAT3, Auranofin.
 Global reach, higher impact
Global reach, higher impact