Impact Factor
Theranostics 2020; 10(18):8018-8035. doi:10.7150/thno.42943 This issue Cite
Research Paper
Specific ablation of CD4+ T-cells promotes heart regeneration in juvenile mice
1. Department of Chemical Pathology; and Li Ka Shing Institute of Health Sciences, Prince of Wales Hospital, The Chinese University of Hong Kong, Hong Kong, China.
2. The State Key Laboratory of Cell Biology, CAS Center for Excellence in Molecular Cell Science, Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences, University of Chinese Academy of Sciences, Shanghai, China.
3. Department of Cardiology, Center for Translational Medicine, Institute of Precision Medicine, The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, China.
4. Lead Contact.
* These authors contributed equally to this work.
Abstract
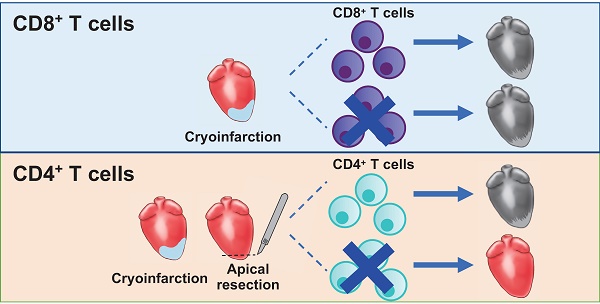
Unlike adult cardiomyocytes, neonatal cardiomyocytes can readily proliferate that contributes to a transient regenerative potential after myocardial injury in mice. We have recently reported that CD4+ regulatory T-cells promote this process; however, the role of other CD4+ T-cell subsets as well as CD8+ T-cells in postnatal heart regeneration has been less studied. Methods: by comparing the regenerating postnatal day (P) 3 and the non-regenerating P8 heart after injury, we revealed the heterogeneity of CD4+ and CD8+ T-cells in the myocardium through single cell analysis. We also specifically ablated CD4+ and CD8+ T-cells using the lytic anti-CD4 and -CD8 monoclonal antibodies, respectively, in juvenile mice at P8 after myocardial injury. Results: we observe significantly more CD4+FOXP3- conventional T-cells in the P8 heart when compared to that of the P3 heart within a week after injury. Surprisingly, such a difference is not seen in CD8+ T-cells that appear to have no function as their depletion does not reactivate heart regeneration. On the other hand, specific ablation of CD4+ T-cells contributes to mitigated cardiac fibrosis and increased cardiomyocyte proliferation after injury in juvenile mice. Single-cell transcriptomic profiling reveals a pro-fibrotic CD4+ T-cell subset in the P8 but not P3 heart. Moreover, there are likely more Th1 and Th17 cells in the P8 than P3 heart. We further demonstrate that cytokines of Th1 and Th17 cells can directly reduce the proliferation and increase the apoptosis of neonatal cardiomyocytes. Moreover, ablation of CD4+ T-cells can directly or indirectly facilitate the polarization of macrophages away from the pro-fibrotic M2-like signature in the juvenile heart. Nevertheless, ablation of CD4+ T-cells alone does not offer the same protection in the adult heart after myocardial infarction, suggesting a developmental change of immune cells including CD4+ T-cells in the regulation of age-related mammalian heart repair. Conclusions: our results demonstrate that ablation of CD4+ but not CD8+ T-cells promotes heart regeneration in juvenile mice; and CD4+ T-cells play a distinct role in the regulation of heart regeneration and repair during development.
Keywords: CD4+ T-cells, neonatal heart regeneration, juvenile heart regeneration, cardiomyocyte proliferation, cardiac fibrosis, macrophages.
 Global reach, higher impact
Global reach, higher impact