Impact Factor
Theranostics 2020; 10(18):8061-8079. doi:10.7150/thno.42284 This issue Cite
Review
Gold-based Inorganic Nanohybrids for Nanomedicine Applications
1. Key Laboratory for Organic Electronics and Information Displays & Jiangsu Key Laboratory for Biosensors, Institute of Advanced Materials (IAM), Nanjing University of Posts and Telecommunications, Nanjing, 210023, China.
2. Department of Dermatology, Nanjing Drum Tower Hospital, The Affiliated Hospital of Nanjing University Medical School, Nanjing, 210008, China.
3. i-Lab and Division of Nanobiomedicine, CAS Key Laboratory of Nano-Bio Interface, CAS Center for Excellence in Nanoscience, Suzhou Institute of Nano-Tech and Nano-Bionics, Chinese Academy of Sciences, Suzhou, 215123, China.
4. School of Nano-Tech and Nano-Bionics, University of Science and Technology of China, Hefei, 230026, China.
Received 2019-11-19; Accepted 2020-6-18; Published 2020-7-2
Abstract
Noble metal Au nanoparticles have attracted extensive interests in the past decades, due to their size and morphology dependent localized surface plasmon resonances. Their unique optical property, high chemical stability, good biocompatibility, and easy functionalization make them promising candidates for a variety of biomedical applications, including bioimaging, biosensing, and cancer therapy. With the intention of enhancing their optical response in the near infrared window and endowing them with additional magnetic properties, Au nanoparticles have been integrated with other functional nanomaterials that possess complementary attributes, such as copper chalcogenides and magnetic metal oxides. The as constructed hybrid nanostructures are expected to exhibit unconventional properties compared to their separate building units, due to nanoscale interactions between materials with different physicochemical properties, thus broadening the application scope and enhancing the overall performance of the hybrid nanostructures. In this review, we summarize some recent progresses in the design and synthesis of noble metal Au-based hybrid inorganic nanostructures for nanomedicine applications, and the potential and challenges for their clinical translations.
Keywords: Au nanoparticles, hybrid nanostructures, localized surface plasmon resonances, nanomedicine, theranostics
Introduction
Although huge economic and scientific efforts have been invested in cancer research worldwide, more than 10 million new cancer cases are diagnosed each year, with death toll continues to rise [1-4]. Biomedical nanotechnology, including molecular diagnostic and nanotherapeutics that provide new options for clinical treatment, has recently been shown as a very promising technology to improve cancer patients' treatment outcomes and reduce the socioeconomic burden. Potential clinical applications of nanotechnology can be categorized depending on its usage on the body as either outside (such as “point-of-care” testing) or inside (such as diagnostics and targeted drug delivery). When focusing on their clinical application inside the body, it is highly desirable to develop strategies that enable real-time tracking of the therapeutic response during the treatment, in order to avoid insufficient treatment or over-dosing side effect. In recent years, theranostic strategies based on various nanomaterials, noble metal Au nanoparticles (NPs) in particular, have received tremendous attention [5-14]. Under AC electromagnetic field (light), free charge carriers in Au NPs are driven into collective oscillations, displaying unique optical phenomena termed as localized surface plasmon resonance (LSPR). This intense light-matter interaction enables huge enhancement of local electromagnetic field, and has been widely exploited in the fields of optical imaging, sensing, and photocatalysis [15-21]. Generally speaking, LSPR can be tuned over a wide spectral window from visible to the near infrared (NIR) region, depending on the NPs physicochemical properties such as size and morphology. Therefore, methods and protocols have been developed in different research labs for synthesizing Au NPs in a variety of shapes, such as nanosphere [22-24], nanocube [25], nanotriangle [26], nanocage [27], and nanoshell [28, 29]. By changing their size (1 to 100 nm), shape, and structure (single particle, alloy, heterodimer, core-shell, etc.), Au nanostructures can display unique linear and nonlinear optical behaviors, enabling their use as strong photosensitizers in phototherapy and in vitro diagnostics [27, 30-34]. Moreover, Au NPs possess large surface areas that can be conveniently functionalized with various biomolecules by means of Au-thiolate chemistry, facilitating the attachment of different moieties, such as antibodies, peptides, and biocompatible polymers with good biocompatibility and targeting capability [35, 36]. The development of facile synthesis and surface functionalization strategies of Au NPs have pushed forward their practical applications in the field of nano-biomedicine, including bioimaging [37-41], drug delivery [42-44], cancer diagnosis, and therapeutics [45-50].
Hybrid nanostructures composed of multiple domains with different compositions have attracted great interests in diverse research fields. For biomedical applications, hybrid nanostructures can provide multimodal imaging modality or imaging-therapy capability all-in-one single unit. More specifically, since Au possess excellent X-ray attenuation ability and high photothermal transduction efficiency, combining Au NPs with metal oxides or metal chalcogenides would either provide complementary imaging modality for accurate cancer diagnosis or offer additional therapeutic avenue for enhanced cancer treatment, thus overcoming the limitation of single theranostic model. Hence, the combined characteristics of Au-based nanostructures would be extremely valuable for their potential applications in precision nanomedicine. Moreover, the construction of plasmonic Au NPs based hybrid nanocomposites may effectively incorporate light absorption, magnetic response, and thermal effect in one single nanostructure. The mutual interaction between Au NPs and neighboring nanomaterials at the nanoscale contact can generate complex interfacial behaviors, such as electron transfer and near-field enhancement, which may induce changes in the effective carrier concentration and optical resonances [51-54]. This plasmon-driven carrier density change and near-field effect in nanohybrids can lead to potential synergistic performance enhancement when compared to the simple sum of the isolated individual components. For example, it is demonstrated that Au NPs can activate the adjacent semiconductors or metal species, enabling increased photoenergy conversion or enhanced light-absorption properties, thus promoting reactive oxygen species (ROS) generation, photoacoustic signal amplification, and heat generation [55-57], benefiting the biomedical outcomes of photodynamic therapy (PDT), photoacoustic (PA) imaging, and photothermal therapy (PTT). Therefore, designing Au-based nanohybrids is a desirable strategy to achieve enhanced theranostic efficiency without increasing the dose of NPs applied, thus averting potential side effect [58-66]. These promising features together with their ease of surface modification make noble metal Au-based nanocomposites a powerful platform for diverse biomedical applications [67-73].
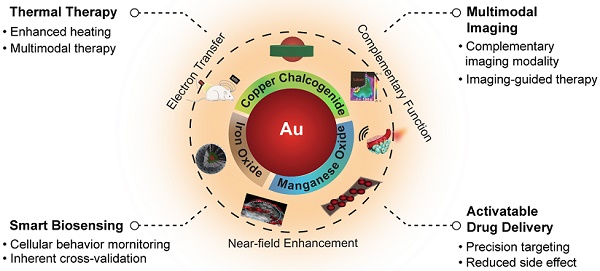
Some excellent reviews have summarized the advances of using noble metal NPs in the field of nanomedicine such as drug delivery, phototherapy, and biosensing [74-76]. However, reviews focusing specifically on Au NPs-based inorganic hybrid nanostructures for biomedical applications are still rare. In this review, we will focus on the design and synthesis of Au-based inorganic hybrid nanostructures, and their improved performance when being applied in the field of nanomedicine, such as bioimaging, cancer therapy, and drug delivery [77-81]. For the choice of adjoining components to Au, we limit our selection to copper chalcogenide, iron oxide, and manganese oxide, which are bioactive nanomaterials that can provide complementary theranostic potential to Au (as schematically illustrated in Figure 1). For each type of nanohybrid, a few important aspects will be discussed including the design and preparation of hybrid nanostructures, interaction between noble metal Au and the adjoining components, as well as their biomedical performance as theranostic agents (as briefed in Table 1).
Illustration of various Au-based inorganic hybrid nanocomposites for diagnostic and therapeutic nanomedicine applications.
Summary of Au-based inorganic hybrid nanostructures used in nanomedicine
| Hybrid structure | Synthetic method | Size | Application | Advantage | Ref. |
|---|---|---|---|---|---|
| Au-Cu9S5 UFO-shape | Seeded growth method | ~22 nm (TEM) | PTT-CT | Improved heating effect | 77 |
| Au-Cu2-xSe heterodimer | Seeded growth method | ~10 nm (TEM) | PA | Deep tissue imaging up to 17 mm | 78 |
| Au-Cu2-xS core-shell | Anion exchange | 85.87±10 nm (DLS) | SERS/PA-PTT | Bimodal imaging-guided PTT | 73 |
| Au-Cu9S5@MSN | Seeded growth method | 107 nm (DLS) | PTT-MRI | MRI tacking drug release | 80 |
| CuS@Cu2S@Au hollow structure | Template method | 100 nm (TEM) | PTT-chemotherapy | Photo-switchable targeting | 96 |
| Au-Cu2-xS/Se core-shell or heterodimer | Seeded growth method | Tunable (TEM) | N.A | N.A | 102 |
| Au@Cu2-xS/Se core-shell | Self-assembly | Tunable (TEM) | PTT-PA-CT | High photothermal conversion efficiency | 103 |
| Cubic CuS@spicky Au core-shell | Template method | 78±5 nm (DLS) | PTT-SERS | Enhanced PTT and SERS | 109 |
| Au-CuS yolk-shell structure | Template method and anion exchange | Tunable (TEM) | PTT-PDT-chemotherapy | Enhanced PTT and PDT | 110 |
| Au-Cu2-xS core-shell | Template method | 150 nm (TEM) | Chemo-PTT | Enhanced photothermal effect | 111 |
| Fe3O4@Au core-shell | Seeded growth method | 6.3 ± 0.7 nm (TEM) | Hyperthermia | Improved hyperthermia | 121 |
| Fe3O4/Au cluster/shell | Seeded growth method | 240 nm (TEM) | SERS-magnetic hyperthermia | Improved hyperthermia | 122 |
| Fe@Au bi-layer semi-shell | Nanolithography and physical vapor deposition | 40 nm (TEM) | CT, MRI, and fluorescence | Magnetically amplified photothermal therapy | 123 |
| Fe3O4/Au cluster/shell | Seeded growth method | 126±11 nm (TEM) | PTT-magnetic hyperthermia | Bimodal thermo-therapy | 124 |
| Au-Fe3O4 heterodimer | Seeded growth method | 11-14 nm (TEM) | MRI-CT | Bimodal imaging | 136 |
| Fe3O4@Au core-shell | One-pot hydrothermal | 262.7±3.06 nm (DLS) | MRI-CT | Bimodal imaging | 137 |
| Fe2O3@Au core-shell | Seeded growth method | 22.1±1.9 nm (TEM) | MRI-CT | Bimodal imaging | 138 |
| Fe3O4@SiO2@Au core-shell | Seeded growth method | 222±1.5 nm (DLS) | MRI/CT(PA) imaging | Bimodal imaging | 140 |
| Fe2O3@Au core-shell | Seeded growth method | 179 nm (DLS) | SERS-PA-MRI-PTT | Tri-modal imaging-guided PTT | 141 |
| Fe3O4@Au yolk-shell | Seeded growth method | 65 nm (TEM) | MRI-PA-PET-chemo-thermal therapy | Multimodal imaging-guided chemo-thermal therapy | 142 |
| Fe3O4@Au@mSiO2 core-shell | Seeded growth method | 10.4 ± 2.3 nm (DLS) | PTT-PDT | Enhanced PDT | 143 |
| Au-Fe3O4 heterodimer | Seeded growth method | 16.7 ± 2.3 nm (TEM) | X-ray protection and X-ray enhancing agents | Discriminate healthy cell and cancer cell | 144 |
| MSN-Au-Fe3O4 core-shell | Assembly | 140 nm (TEM) | Nanozyme | Nanozyme-catalyzed cascade reaction | 145 |
| Au@MnO2 core-shell | Seeded growth method | 50 nm (TEM) | Radiotherapy | Overcoming the hypoxia-associated radiotherapy resistance | 158 |
| Au cage@MnO2 core-shell | Seeded growth method | 91 nm (TEM) | PDT | Boost immunogenic PDT | 159 |
| Cu2-xSe (Au)@MnO2 core-shell | Seeded growth method | 60 nm (TEM) | PTT | Redox-activated MRI-guided PTT | 162 |
| Au@MnO2 UFO-shaped | Seeded growth method | 230 nm (TEM) | Dark field imaging | Monitoring cell membrane vesiculation | 163 |
| Au@MnO2 core-shell | Bio-templated method | 20-25 nm (TEM) | Fluorometric and MRI based sensing | Inherent cross-validation | 164 |
Abbreviations: PTT: photothermal therapy; PDT: photodynamic therapy; PA: photoacoustic imaging; CT: computed tomography; MRI: magnetic resonance imaging; SERS: surface enhanced Raman scattering; MSN: mesoporous silica nanoparticle; TEM: transmission electron microscopy; DLS: dynamic light scattering.
Au-Cu2-xE nanocomposites in nanomedicine
Other than the most researched noble metal nanocrystals, recent studies find that heavily-doped semiconductor nanocrystals such as non-stoichiometric copper chalcogenide NPs (Cu2-xE, where E = S, Se, Te; 0<x≤1) with different compositions can also support LSPR, due to their positively charged free carriers [82-86]. This opens up a new field for plasmonic research [87-94], as LSPR of semiconductor nanocrystals can be easily tuned from visible to NIR by simply changing their doping levels. For biomedical applications, the emergence of copper chalcogenide nanocrystals circumvents the limitations experienced when using NIR absorbing anisotropic Au nanocrystals, which are generally large in size and unstable under laser irradiation conditions. Moreover, combining the traditional plasmonic noble metal Au with copper chalcogenide has attracted increasing attention in recent years. Many research groups have devoted efforts into constructing dual plasmonic noble metal-doped semiconductor nanocrystal hybrids, and investigated their coupled surface plasmon resonance properties and applications in the fields of catalysis and nanomedicine [77, 95, 96].
Photothermal therapy
By changing the doping levels either chemically or electrochemically, the LSPR of Cu2-xE can be tuned dynamically, showing characteristic LSPR peaks extendable to the second NIR (NIR-II) window (1000-1350 nm), which is the optimal biological transparent window with larger optical penetration depth and higher maximum permissible exposure of light irradiation over the traditional NIR-I window (700-950 nm) [97-101]. Through the construction of dual plasmonic nanohybrids, the LSPR coupling between Au and Cu2-xE may open up a new regime for designing photo-absorbers with enhanced photothermal efficiency, an attractive attribute for imaging and therapy applications in the NIR-II window.
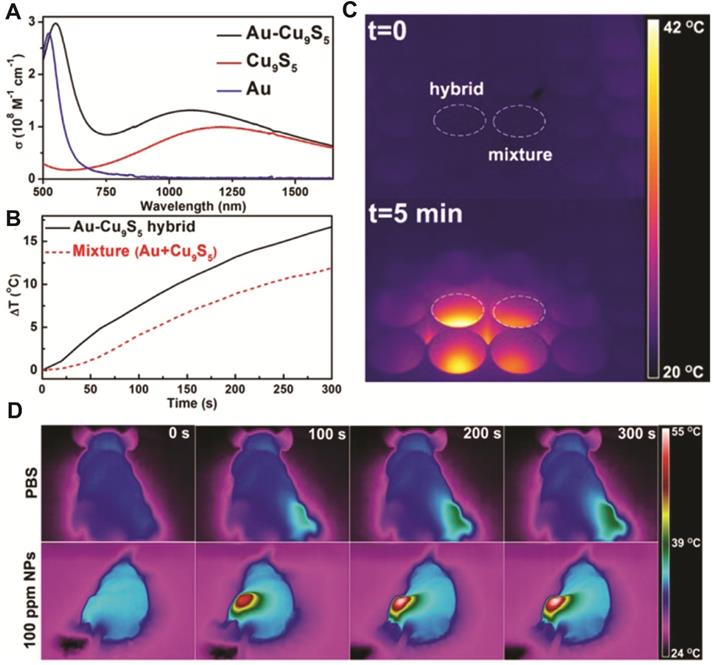
In 2014, our group constructed a dual plasmonic hybrid Au-Cu9S5 with well-controlled interfaces [77]. Using the high purity heterodimer nanohybrid, we investigated the LSPR coupling effect originating from the collective electron and hole oscillations in the hybrid system, and found that the synergistic interactions between two components contributed to their enhanced photothermal performance in the NIR-II window. When comparing the molar extinction coefficient of the hybrid NP to that of its individual components (Au and Cu9S5), the Au-Cu9S5 hybrid showed a 50% enhanced absorption at 1064 nm compared to pure Cu9S5 NPs synthesized using the same protocol (Figure 2A). This enhanced NIR absorption further translated to improved heating capability (Figure 2B), as shown clearly in the thermal images (Figure 2C). The light penetration depth in the NIR-II window was also explored and a decay length of 5.3 mm at 1064 nm was determined. The experimentally measured photothermal performance and theoretical calculations revealed strong LSPR interaction between Au and Cu9S5 domains in the nanohybrids. When being used for in vivo photothermal therapy, more than 10 °C increase was observed at tumor site under 1064 nm laser irradiation at a power density of 0.6 W cm-2, which is higher than the required effective temperature for cancer photothermal therapy (42-45 °C), thus inducing significant tumor ablation (Figure 2D). By combining X-ray computed tomography (CT) imaging and photothermal therapy capabilities in one nanostructure, the Au-Cu9S5 nanohybrids were demonstrated to be an attractive multifunctional platform for theranostic application. As the first report on efficient photothermal therapy in the NIR-II windows with power density lower than laser safety standards (1 W cm-2, ANSI Z136.1-2007, American National Standard for Safe Use of Lasers), this work reveals that constructing dual plasmonic nanostructures and optimizing the coupling effect of LSPR in nanohybrids is an efficient strategy to design better-performing theranostic agent in the NIR-II window.
(A) Molar extinction coefficient of Au-Cu9S5 hybrid nanostructures and corresponding Au and Cu9S5 NPs. (B) Temperature increment of Au-Cu9S5 hybrid nanostructures compared to the physical mixture of Au and Cu9S5 NPs at the same concentrations. (C) Comparison of temperature changes captured by a thermal imaging camera from Au-Cu9S5 hybrids and the physical mixture of Au and Cu9S5 NPs under laser irradiation. (D) Representative thermal images of tumor-bearing mice under the irradiation of 1064 nm laser (0.6 W cm-2). Images are reproduced with permission from [77], copyright 2014 American Chemical Society.
Following the work of Au-Cu9S5 nanohybrids construction, various synthetic methods have been deployed to integrate Au in different shapes with copper chalcogenides to form dual plasmonic nanostructures of tunable geometries, in order to explore their LSPR coupling effect and enhanced photothermal capacity. To establish a more general strategy for synthesizing dual plasmonic nanocomposites, our group developed a facile aqueous phase synthesis route to integrate plasmonic Au with self-doped semiconductor Cu2-xSe [102]. Using a Se-mediated approach, Au-Cu2-xSe hybrid nanocrystals with different Au core morphologies such as nanoparticle, nanorod, and nanotriangle can be facilely synthesized. Moreover, Au-Cu2-xSe hybrid nanocrystals with different morphologies such as core-shell and heterodimer geometry can be obtained by varying the polymers used for nanocrystal stabilization. Independently, Xia and coworkers developed a general and eco-friendly method to synthesize core@shell Au@Cu2-xE (E = S, Se) dual plasmonic nanohybrids in aqueous solution for multimodal imaging and tumor therapy applications [103]. Due to the plasmonic coupling between noble metal core and semiconductor nanoshell, the as-prepared hybrid Au@Cu2-xS showed an extremely large extinction coefficient of 9.32 L g-1 cm-1 at 808 nm. Another approach for obtaining Au@Cu2-xS core@shell NPs with independently tunable core and shell morphology was developed by Zhang et al., through a cation exchange enabled non-epitaxial strategy [95], where the nonstoichiometric composition and thickness of the Cu2-xS shell can be precisely controlled.
Photoacoustic imaging
Photoacoustic (PA) imaging modality is based on measuring the acoustic waves generated in biological tissues after short laser pulses excitation [104-106]. By combining the advantage of high spatial resolution from optical imaging and large penetration depth from ultrasound detection, PA imaging has become a fast-developing imaging technique with great potential in biomedical and clinical applications [41]. PA imaging contrast depends on the optical cross sections of the tissue and the injected imaging agents. Therefore, strongly absorbing plasmonic nanocrystals including Au and copper chalcogenides have been selected as candidates for PA imaging contrast enhancing agents [107-109].
Based on the plasmonic coupling induced enhanced photothermal response of Au-copper chalcogenides nanohybrids, Swihart and coworkers have reported using Au-Cu2-xSe heterodimer nanocrystals as contrast agents for deep tissue PA imaging [78]. The Au-Cu2-xSe heterodimer NPs exhibited a broad optical absorption spectrum across both NIR-I and NIR-II window, as a result of electron transfer between the constituting Au and Cu2-xSe domains (Figure 3A). Under 1064 nm excitation, with a power density (10 mJ cm-2) at only 1/10 of the ANSI safe limit, sentinel lymph node (SLN) mapping up to 17 mm under skin was achieved (Figure 3B), demonstrating their potential for clinical applications.
(A) UV-vis spectra of Au-Cu2-xSe heterodimers and the 4.6 nm Au seed NPs. (B) Representative PA imaging of sentinel lymph node before (b1) and after Au-Cu2-xSe injection for 68 min (b2) and 251 min (b3), and the depth-encoded PA coronal image (b4). Images are reproduced with permission from [78], copyright 2013 American Chemical Society. (C) Schematic illustration of endogenous H2S-triggered enhanced PA imaging and photothermal therapy based on LSPR coupling effect. (D) In situ sulfidation of Au@Cu2O nanocomposites and the corresponding PA images before and after intratumoral injection of Au@Cu2O nanostructures at different time points. Images are reproduced with permission from [81], copyright 2019 John Wiley and Sons.
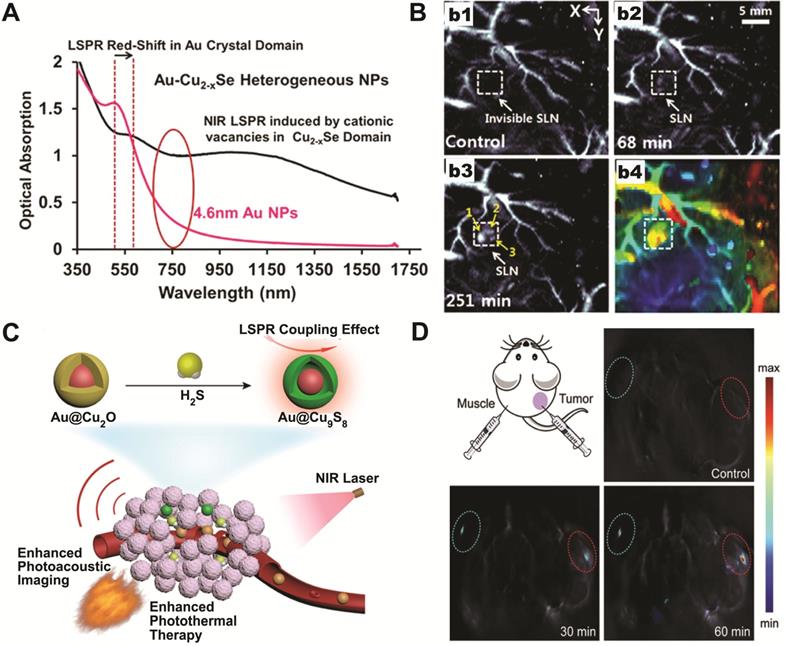
As photothermal and photoacoustic effects are intrinsically related to the light-matter interaction, the dual plasmonic Au-copper chalcogenides nanohybrids are perfect candidates for PA imaging-guided photothermal therapeutic applications operating at the same NIR window. Nie et al. reported aqueous phase synthesis of Au@Cu2-xS core-shell NPs via anion exchange between S2- and Au@Cu2O core-shell NPs, which were then used for accurate tumor identification and efficient ablation through PA imaging-guided photothermal therapy (Figure 3C) [73]. The idea of chemical conversion from Cu2O to CuS using S2- was further utilized for smart theranostic agent design [81]. A characteristic physiological feature of colon cancer is the high level of endogenous hydrogen sulfide (H2S). Yang and coworkers have shown that the photothermal conversion efficiency of Au@Cu2O increased 50% in the presence of NaHS. Moreover, after intratumoral or intravenous injection, in situ sulfidation of Au@Cu2O by endogenous H2S in colon tumor was confirmed by both photoacoustic imaging (Figure 3D) and Raman spectroscopy. The converted Au@Cu9S8 showed about twice stronger absorption at 808 nm, with increased photothermal conversion efficiency ~1.2 times higher than the original Au@Cu2O. This work demonstrates that the in situ generated Au-copper chalcogenides nanohybrids, formed by responding to local physiological niche environment at tumor site, can act as smart PA imaging-guided photothermal theranostic agent to treat cancers.
Activatable drug delivery
Photothermal therapy can be used to eradicate tumor cells through localized heating. However, unsatisfactory tumor inhibition may occur due to inhomogeneous heating effect at tumor site. Combining chemotherapy with photothermal therapy has shown great promise in cancer treatment, where local heating can be used to regulate drug release with both spatial and dosage control, while the elevated local temperature also improves drug efficacy in treating cancer. To enable higher drug loading capacity, voids are often introduced into the nanohybrid design. For instance, Lin et al. described the synthesis of hollow CuS@Cu2S@Au nanostructures, which not only exhibited enhanced photothermal conversion efficiency, but also afforded high drug loading capability by providing large cavity and mesoporous shell, thus enabling photo-responsive drug release under NIR laser excitation [96]. Zhang's group developed Au@void@CuS yolk-shell nanostructures as multifunctional drug carriers [110]. After doxorubicin (DOX) loading, the resultant DOX-Au-CuS yolk-shell nanocomposites could kill cancer cells more efficiently than the unloaded NPs under the same 980 nm laser irradiation conditions, due to the simultaneous photothermal and chemotherapeutic effect.
Drug delivery vehicles are often responding only to single stimulus such as external light irradiation or endogenous pH environment. Designing drug delivery system responding to multiple stimuli will not only minimize the undesirable release of chemotherapeutics thus avoiding adverse side effects, but also maximize the drug dosage in the target region with improved drug availability. To reach this goal, Cao et al. designed a dual responsive drug release system utilizing a rattle-type Au@Cu2-xS hollow mesoporous structure [111]. With this structure, a high drug loading capacity of 908 μg DOX per mg of the hollow mesoporous nanocrystals was achieved. More importantly, the obtained hybrid nanostructures displayed both endogenous pH- and external photo-responsive drug release behaviors. The acidic pH mimicking tumor microenvironment and NIR laser irradiation could activate the drug delivery system with over 70% of DOX release in 20 min. This makes Au@Cu2-xS hollow mesoporous structure a promising agent for chemo-photothermal therapy under photoacoustic imaging guidance, due to their superb photothermal conversion efficiency and conspicuous capability of photo activatable drug release property.
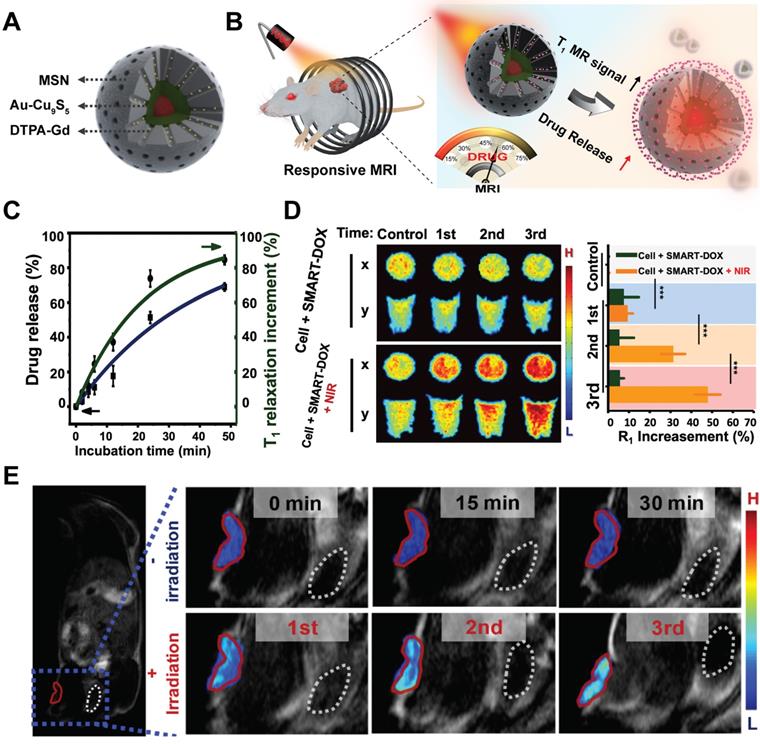
Further integrating activatable drug delivery system with real-time drug release monitoring function would enable patient-specific drug administration and benefit personalized medical treatment. Recently, based on the Au-Cu9S5 nanostructures, we have developed a smart drug delivery platform with noninvasive activatable magnetic resonance (MR) imaging capacity for controllable drug release tracking [80]. The smart nanocomposites contained two functional components, which were the inner Au-Cu9S5 core for heat generation under laser irradiation and outer mesoporous silica (MSN) shell for drug molecules loading and paramagnetic Gd3+ ions anchoring (Figure 4A). The paramagnetic Gd3+ ions-based chelates were used clinically to accelerate the longitudinal relaxation (T1) of excited protons, thus increasing the longitudinal relaxation rate (r1) and generating enhanced T1 MR images. Under exogenous NIR-II irradiation, localized heating of Au-Cu9S5 core would melt the gatekeeper phase-change materials loaded in the MSN shell and trigger pulsated drug release with good on/off control (Figure 4B). With the released drug molecules leaving the hybrid nanocomposites, the accessibility of proton to the paramagnetic Gd3+ ions anchored in the mesoporous channels was significantly promoted, which improved r1 of protons, and resulting in a positive correlation between T1 MR imaging signal and the amount of released drugs (Figure 4C), which was further verified at cellular (Figure 4D) and in vivo levels (Figure 4E).
Schematic illustration of Au-Cu9S5@MSN nanostructures (A) and their NIR responsive drug release behavior with real time MRI monitoring property (B). (C) DOX release from Au-Cu9S5@MSN-DOX nanocomposites at 45 °C, and the corresponding T1 relaxation increment. (D) Evolving T1-weighted MR images and MR relaxations of cancer cells treated with Au-Cu9S5-DOX nanocomposites after different repetition of NIR irradiations. (E) T1-weighted MR images of mice injected with Au-Cu9S5-DOX nanocomposites with and without laser irradiation treatments. Images are reproduced with permission from [80], copyright 2019 Springer Nature.
Au-FexO nanocomposites in nanomedicine
Iron oxide (Fe2O3 and Fe3O4) with good chemical stability and biocompatibility has gained tremendous attention in diverse biomedical applications including magnetic resonance imaging, sensing, remote-controlled drug delivery, and magnetic hyperthermia [112-119]. By integrating magnetic iron oxide and plasmonic Au into one single unit, the as formed magneto-plasmonic hybrid nanostructures possess great potential in theranostic applications. Their optical and magnetic properties can be tuned independently by changing the respective nanoparticle domain size, shape, composition, and geometry. The unique attributes of magneto-plasmonic hybrid nanostructures have attracted great interests into their design and application for simultaneous diagnosis and treatment of cancer.
Thermal therapy
Thermal therapy is a promising approach to kill cancer cell with the local temperature at tumor site reaching 42-45 °C. Iron oxide and Au NPs are typical thermal agents that can generate heat to destroy tumor cells through noninvasive interaction with either oscillating magnetic field or NIR light [120]. However, both magnetic hyperthermia and photothermia have their inherent drawbacks. While noble metal Au-based photothermia show high heating efficiency with good spatial resolution, the compromised light penetration depth in living tissues set a limit on its potential clinical applications. On the other hand, magnetic NP mediated hyperthermia employs radiofrequency, thus overcoming the penetration depth limitation of photothermia. Unfortunately, magnetic hyperthermia utilizing biocompatible iron oxide NPs suffers from their low specific loss powers. Extensive efforts have been put into modulating the size, magnetization or anisotropy of magnetic particles to enhance their specific absorption rate, thereby improving their heat generation capacity. Designing magneto-plasmonic nanostructures through hybridizing noble metal Au with magnetic nanomaterials together has been explored to overcome the limitations set by the individual components. It was found that by capping magnetic NPs with Au, more local heat could be generated when the hybrids were put under a radiofrequency field. Challa S. S. R. Kumar et al. demonstrated that superparamagnetic iron oxide (Fe3O4) NPs (SPION, 5.4 nm) coated with 0.4-0.5 nm thick gold nanoshell can generate 4-5 times more heat compared to that of the pure Fe3O4 NPs under a low-frequency oscillating magnetic field [121]. They speculated that the higher heat generation capacity was attributed to larger magnetic anisotropy of the superparamagnetic Fe3O4 NPs inside Au shell. Zhou's group reported a similar study recently [122]. Under the same magnetic induction conditions, a local temperature of 15 °C higher was achieved with the hybrid Fe3O4/Au cluster/shell nanostructures compared to that of the pure Fe3O4 NPs, and induced higher percentage of cancer cell apoptosis. Furthermore, the Fe3O4/Au nanostructures possessed high transverse relaxation rate (r2) for MR imaging (MRI), while the Au nanoshells can be used as surface enhanced Raman scattering (SERS) substrate for early diagnosis. SERS is a surface enhanced optical phenomenon, as Raman signals from surface-absorbed molecules are significantly amplified, due to the strongly enhanced near-field on the surface of noble metal NPs as the result of LSPR excitation. SERS allows optical sensing with high spatial resolution and sensitivity down to single molecule level under optimal conditions. Other than improving magnetic hyperthermia, photothermal effect can also be magnetically amplified via magnetophoretic manipulation strategy, as illustrate in the work by Sepúlveda et al. [123], where the optical heating efficiency of the Fe/Au nanodomes could be dramatically enhanced by local NPs enrichment in the laser irradiation zone under the assistance of an external magnetic field. In addition to single hyperthermia modality, magnetic hyperthermia and photothermia can be synergistically integrated in a properly designed magneto-plasmonic nanohybrid. Abou-Hassan and coworkers synthesized Au nanoshell coated iron oxide multi-core magneto-plasmonic nanohybrids with diameter of around 30 nm [124]. They demonstrated that the heat generated by the magneto-plasmonic nanohybrids display a cumulative effect when both magnetic and plasmonic heating modalities are working simultaneously. With the treatment dose only 1/10 of that used in typical magnetic hyperthermia therapy, a rapid temperature increase to 48 °C could be achieved in tumor tissue under simultaneous magnetic induction and laser irradiation treatment.
Multimodal MR/(CT, PA, SERS) Imaging
Biomedical imaging is important for early diagnosis and treatment evaluation, which has emerged as a key technology for the development of targeted therapies. Combining multiple imaging tools together can be very helpful in personal and precision medicine. While molecular imaging tools such as positron emission tomography (PET), computed tomography (CT), and FO (fluorescence optical) imaging have been widely used in clinical diagnostics, each of these imaging methods possesses its own strengths and weaknesses. In recent years, integrating different imaging modalities together by designing multimodal imaging agents such as CT/MR, FO/MR, PA/MR, and SERS/MR have been suggested to obtain more comprehensive pictures for accurate cancer diagnosis [125-132]. By integrating magnetic nanomaterials with Au, the concomitant MR imaging modality can provide non-invasive imaging, large penetration depth, and good soft tissue contrast.
Hybrid Au-FexO nanocomposites are considered potential bimodal CT/MR imaging agents [133-135], where FexO component serves as T1 or T2 MR contrast enhancer, while Au with efficient X-ray attenuation capability works as powerful CT contrast enhancing agent. Gu et al. fabricated Au-Fe3O4 heterostructures for bimodal MR/CT imaging application by a seeded-growth method [136]. The prepared Au-Fe3O4 heterostructures were composed of 14 nm Fe3O4 attached to 11 nm spherical Au NPs. The r2 value of the heterostructures was determined to be 136.4 mM-1 s-1 at 1.5 T. Using a rabbit model, the Au-Fe3O4 heterostructure composites exhibited excellent MR/CT contrast enhancing performance. The rabbit liver can be clearly observed by MR imaging. Meanwhile, the detailed anatomical structures such as rabbit right ventricle can be clearly viewed by CT imaging. Using a facile one-pot strategy, Shi et al. reported core-shell Fe3O4@Au nanostructures for bimodal MR/CT imaging application [137]. The MR and CT performance evaluation showed that the hybrid NPs possess high r2 relaxivity (146.07 mM-1 s-1) and excellent X-ray attenuation ability, which was then successfully applied to aorta CT imaging and liver MR imaging in mouse models. In another study, the same group fabricated Fe3O4/Au nanocomposites based on a layer-by-layer (LBL) strategy [135]. Their results demonstrated that at the optimized molar ratio of Au to Fe3O4, Fe3O4/Au NPs exhibited excellent X-ray attenuation characteristics and a relatively high r2 relaxation rate of 92.67 mM-1 s-1. By further modifying them with targeting molecule folic acid, the hybrid nanocomposites could be specifically uptaken by cancer cells that over express folic acid receptors on cell membrane surface. Similarly, Zhang and Wang's group reported the use of lectin conjugated Fe2O3@Au as bimodal MR/CT imaging agent in vivo, targeting specifically the colorectal cancer [138].
Besides attenuating X-rays for CT imaging, Au is also excellent PA imaging agent due to their LSPR characteristics. The construction of bimodal MR and PA molecular imaging agents can overcome the limitation of finite penetration depth of PA imaging, and provide structural and functional information of disease with high resolution and sensitivity. Melancon et al. fabricated multifunctional superparamagnetic Fe3O4@Au nanoshells with excellent PA imaging performance and high r2 relaxivity of 208 mM-1 s-1 [139]. Based on the high NIR absorption and strong magnetic properties of Fe3O4@Au nanoshells, the hybrid Fe3O4@Au nanoshells were capable of lighting up tumor region with PA-MR imaging. Moreover, the bimodal PA and MR imaging can be used to monitor the therapeutic treatment outcome mediated by the photothermal effect of Fe3O4@Au. Functionalizing Au-FexO nanocomposites with targeting ligand could further improve their diagnostic capability. Franchini et al. synthesized a multilayered Fe3O4@SiO2@Au core-shell nanostructure conjugated with folic acid [140]. With hydrodynamic diameter of 222±1.5 nm, the as-prepared nanostructure showed bimodal MR/PA imaging ability. After systemic injection into a tumor bearing mice, PA imaging revealed that Fe3O4@SiO2@Au had exclusively accumulated in the ovarian cancer region after 4 h. These studies demonstrate the great potential of utilizing Au-FexO hybrid nanostructures as bimodal MR/CT(PA) imaging agent for in vivo diagnostic applications. It is noteworthy that the biomedical imaging performance of hybrid nanostructures is strongly associated with their specific geometric arrangement. While both Au-Fe3O4 heterostructure and Fe3O4@Au core-shell nanostructure can be used for bimodal MR/PA imaging, the core-shell Fe3O4@Au nanostructures with LSPR located in the NIR region are obviously more suitable for biomedical MR/PA imaging.
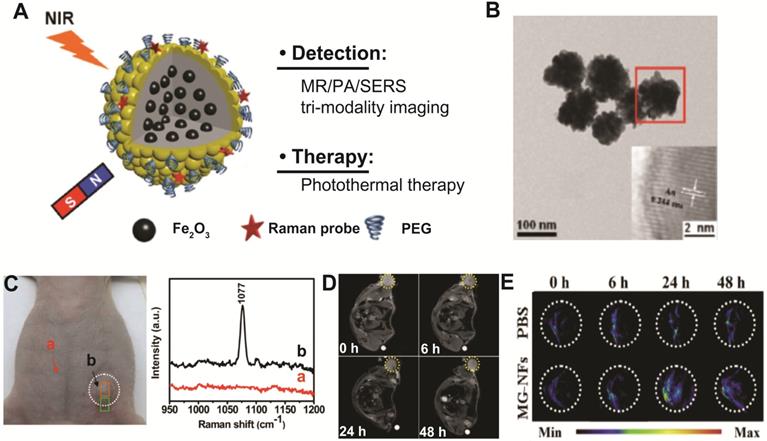
Zhang's group has investigated the potential of using Au-FexO hybrid nanocomposites as multimodal SERS/PA/MR imaging-guided photothermal therapeutics by designing Fe2O3@Au core-shell structure (Figure 5A-B) [141]. The combined tri-modal imaging modality (SERS/PA/MR) can provide complementary information of anatomical tumor localization and tumor resection margin for accurate tumor diagnosis and surgical treatment guidance (Figure 5C-E). Furthermore, due to strong NIR absorbance derived from Au nanoshell, Fe2O3@Au core-shell nanostructures show high photothermal transduction efficiency for cancer therapy. The 4T1 tumor bearing mice administered with Fe2O3@Au core-shell nanostructures have shown significant photothermal tumor ablation under 808 nm laser irradiation. These results illustrated that rationally designed magneto-plasmonic hybrid nanostructures can be used for efficient multimodal imaging-guided photothermal cancer therapy.
(A) Schematic illustration of Fe2O3@Au core-shell nanoflowers for multimodal imaging-guided tumor therapy. (B) Typical TEM image of Fe2O3@Au nanoflower structures. In vivo SERS spectra (C) from normal tissue (a) and tumor region (b), T2-weighted MR images (D), and PA images (E) of a 4T1 tumor bearing mouse injected with either Fe2O3@Au nanoflowers or PBS. Images are reproduced with permission from [141], copyright 2015 John Wiley and Sons.
Activatable drug delivery
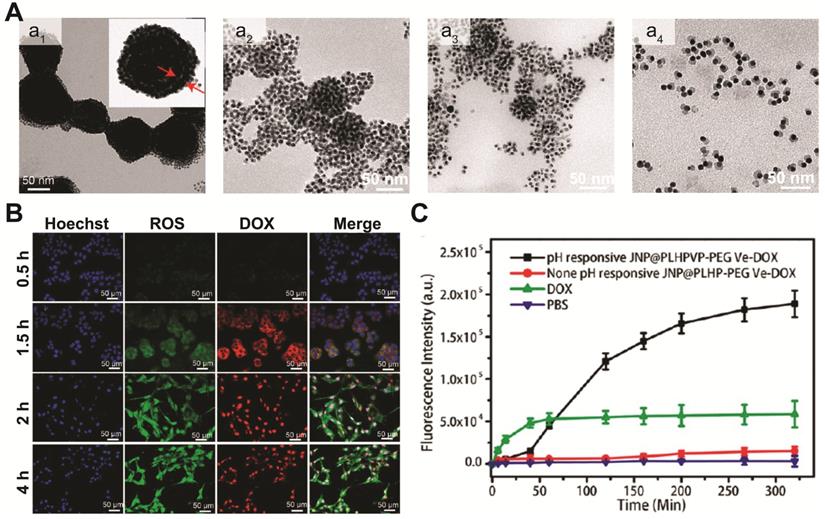
Under external stimulus such as NIR light and magnetic field, the multifunctional Au-FexO hybrid nanostructures can not only act as in vivo diagnostic imaging agent, but also serve as powerful delivery vehicles for controlled drug release. In recent years, efforts have been put into developing different Au-FexO hybrid nanostructures with high drug loading capacity, versatile targeting ability, and smart drug release capability. Chen and coworkers reported the synthesis of a yolk-shell plasmonic-magnetic hybrid theranostic platform [142], which was composed of a small Fe3O4 core encapsulated inside a hollow cavity formed by a porous Au nanoshell. With a relative small size of around 65 nm, the yolk-shell Fe3O4-Au NPs displayed a high r2 value of 149.4 mM-1 s-1, which is ~2.4 times of that from the core-shell structures, indicating that the interfacial interaction of the two components can greatly affect their magnetic properties. In addition, the hollow cavity can be an ideal storehouse for drug loading. After constructing a gatekeeper on the surface using thermosensitive poly(N-isopropylacrylamide-co-acrylamide), thermal responsive drug release is achieved under NIR light irradiation. Initially, only weak DOX fluorescence was observed in the cells as a result of fluorescence quenching by Fe3O4-Au. Upon NIR exposure for 5 min, both cytoplasm and nucleus of the cells displayed strong red fluorescence, suggesting DOX was released, which was also confirmed in the in vivo study. In addition to light trigged drug delivery, pH responsive release system has also been widely adopted in different drug carrier designs. Recently, the same group developed a magnetic-plasmonic bilayer vesicle by assembling Fe3O4-Au janus structure with a pH-responsive polymer for multimodal imaging-guided cancer therapy [79]. The large hollow cavity formed in the assembled bilayer structures enables a high DOX loading capacity. Due to inter-particle plasmonic and magnetic coupling, the assembled bilayer structures displayed enhanced light absorption and high T2 relaxivity, and exhibited improved MRI/PA contrast and photothermal activity, compared to the individual components. Moreover, the bilayer vesicles can be disassembled in mildly acidic microenvironment (Figure 6A). Therefore, DOX loaded in the hollow cavity can be released from the Fe3O4-Au-DOX bilayer vesicles, in response to the decreased pH level in tumor microenvironment (Figure 6B-C). Other than loading chemotherapy drugs, Au-Fe3O4 hybrid nanostructures can also carry singlet oxygen (1O2) photosensitizers, and act as a potential agent for photodynamic therapy. For example, Rosa-Pardo et al. designed a core-shell Fe3O4@Au@mSiO2 nanostructure with photosensitizer Rose Bengal (RB) encapsulated inside mesoporous silica [143]. Due to the surface plasmon sensitization effect of Au shell, a 1.5-fold enhanced 1O2 generation by RB was detected. Furthermore, Au-Fe3O4 nanocomposites are also efficient ROS generating agents with their intrinsic enzyme-mimic characteristics [144-146]. By deliberately designing hybrid nanostructures with multi-enzymatic activities to achieve cascade reactions, high chemo-dynamic therapeutic efficiency has been demonstrated using inorganic nanohybrids. For example, Shi and Chen et al. constructed mesoporous silica coated Au-Fe3O4 nanostructures [145]. In tumor microenvironment, the Au domain behaves as glucose oxidase-mimicking nanozyme, catalyzing glucose to H2O2 and gluconic acid. At the same time, the adjacent Fe3O4 domain acts as peroxidase-mimicking nanozyme, reacting with the in situ generated H2O2 and producing highly toxic ROS to kill cancer cells.
(A) Representative TEM images of assembled Fe3O4-Au janus structures (a1) after being incubated in acidic solution (pH=5.4) for 30 (a2), 60 (a3), and 90 min (a4). (B) Representative fluorescence images of tumor cells incubated with Fe3O4-Au-DOX nanocomposites at different time intervals. (C) Quantification of released DOX by measuring its fluorescence signals. Images are reproduced with permission from [79], copyright 2019 American Chemical Society.
Au-MnO2 nanocomposites in nanomedicine
Besides iron oxides family, manganese oxides such as MnO, MnO2, and Mn3O4 have also been considered as promising candidates for biomedical applications [147-152]. As Mn is one of the essential trace elements in human body, Mn-based nanoparticles such as MnO2 can be utilized and metabolized by the human body. Mn-based complexes are considered as very promising clinical agents for T1 MR imaging. For instance, Mn-dipyridoxyl diphosphate (DPDP) complex Mangafodipir has already been approved as an efficient T1 MR agent for liver imaging. In addition, MnO2 nanomaterials can respond to tumor microenvironment cues such as hypoxia, acidosis, and vascular endothelial growth factor, which can be utilized to amplify their diagnostic and therapeutic performance. For example, MnO2 nanosheets will rapidly decompose and release Mn2+ under mildly acidic and reducing conditions, thus enhancing the contrast of T1 MR imaging [153, 153]. Moreover, the released Mn2+ could initiate Fenton chemistry to kill cancer cells by catalyzing tumor endogenous H2O2 into toxic reactive oxygen species [149]. Therefore, combining MnO2 nanomaterials with the unique LSPR characteristics of noble metal Au can provide a promising theranostic platform as well as form smart probes for versatile biomedical applications both in vitro and in vivo.
Enhanced radiotherapy/photodynamic therapy
Au-MnO2, a new kind of smart therapeutic agent, may serve as a potential theranostic candidate in the field of nanomedicine based on their good biocompatibility and tumor microenvironment responsive behaviors [149, 153-157]. The heavy atom Au can absorb X-rays to generate charged particles, and enhance the effect of radiotherapy (RT). Meanwhile, the MnO2 domain can react with endogenous H2O2 in tumor microenvironment to generate oxygen locally, thus overcoming hypoxia-associated RT resistance. For this purpose, Liu et al. designed Au@MnO2 nanostructures [158], and they observed that Au@MnO2 hybrids indeed have enhanced radiotherapy efficiency as designed. In addition, the nanocomposites containing Au and MnO2 NPs also showed enhanced performance as photodynamic agents [159]. In treating metastatic triple-negative breast cancer, core-shell Au nanocage@MnO2 structures were able to boost immunogenic photodynamic therapy (PDT), thus inhibiting tumor growth and metastases. The enhanced therapeutic efficiency is attributed to the tumor microenvironment responsive oxygen generating MnO2 components, which was decomposed at acidic tumor H2O2-rich conditions and produced sufficient oxygen to boost PDT effect originating from the adjoining Au nanocage.
Responsive imaging
Novel tumor microenvironment-responsive imaging agents have emerged as a promising class of theranostic agent for imaging-guided cancer treatment. By designing tumor microenvironment responsive nanoprobes, large off/on imaging contrast can be achieved. For example, Meng et al. prepared Au nanostar@MnO2 nanosheet hybrid structure of less than 50 nm in dimension [160]. With strong light absorption from 300 to 800 nm, the as synthesized hybrid can destroy tumor cells effectively through photothermal effect. Furthermore, the hybrid nanostructures displayed enhanced MR imaging capability in the presence of GSH, due to their redox environment responsive MR imaging capability. This study demonstrates the potential of Au-MnO2 nanocomposites as efficient theranostic nanoprobes for activatable MR imaging-guided photothermal therapy. Other than photothermal therapy, many other therapeutic modalities such as photodynamic therapy and chemotherapy have also been integrated with MnO2 components. Lin's group designed MnO2-Pt@Au25 nanocomposites, which combined photodynamic therapy, chemotherapy, and activatable MR imaging together in one system [161]. The MnO2 nanosheets acted as carrier for both photosensitizer Au25 and prodrug Pt(IV) loading. In the reducing tumor microenvironment, high level of GSH would be consumed through redox reaction with MnO2 nanosheets and Pt(IV) prodrugs. As a result, both photodynamic therapeutic efficiency induced by Au25 cluster and Pt(II) chemotherapy efficiency were enhanced. More importantly, the reduced Mn(II) ions released from MnO2 nanosheets can increase the MR relaxivity from 401.9 mg-1 s-1 (r1) and 48.8 mg-1 s-1 (r2) to 471.3 mg-1 s-1 (r1) and 49.6 mg-1 s-1 (r2), thus enhancing their corresponding T1 and T2 MR imaging contrasts. Our group has also reported a feasible strategy to decorate various core materials including Au nanoparticle and Au nanorod with the tumor microenvironment-responsive MnO2 shell, which can be utilized as activatable MRI-PTT theranostic platforms for cancer therapy [162].
Smart biosensing platform
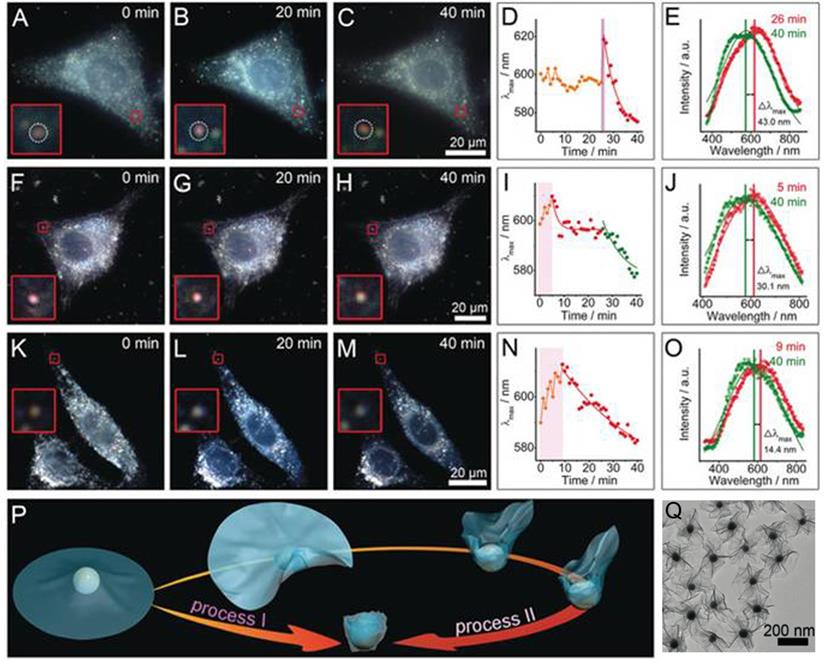
Under a dark-field microscope, a special condenser is used to block central light so that a circular light cone is incident on the object at high angle, only allowing oblique rays to hit the object. This blocks zeroth order light, and objects scatter light more strongly will stand out from the non-scattering dark background. Therefore, Au NPs with strong light-matter interaction due to their LSPR characteristics are perfect objects to be imaged under a dark-field microscope. In this regard, Au-MnO2 nanohybrids are gaining interest as smart biosensors for probing complex cellular events. Xia and coworkers developed UFO-shaped Au-MnO2 plasmonic supraparticles with diameter of around 230 nm, and used these anisotropic structures as dark-field contrast agents to probe the nano-bio interaction at the single cell level (Figure 7A-O) [163]. Due to the flexibility of thin MnO2 nanosheets, they can be physically deformed and folded during the endocytosis process. By employing dark-field spectroscopy, they visualized the interactions between 2D Au-MnO2 nanostructures and living cells, and identified two definitely different trans-membrane processes (Figure 7P). During the cell membrane wrapping process, the deformation and folding of the thin MnO2 nanosheets (Figure 7Q) induced effective refractive index changes around Au NPs, rendering the NPs LSPR scattering red shift with different magnitudes depending on the endocytosis process. On the other hand, the presence of redox species within cells would disintegrate MnO2, and induce a LSPR blue-shift, which could be employed to mark the complete cell membrane engulfment process. This LSPR modulation approach provides a convenient but efficient way to monitor the dynamic interactions between nanomaterials and cells. In addition to serve as a cellular probe, smart Au-MnO2 nanocomposites can also be employed for point-of-care testing. Au@MnO2 hybrid nanocomposites have been developed to detect ascorbic acid (AA) in human serum [164]. The redox reaction between Au@MnO2 nanocomposites and AA resulted in the degradation of MnO2, inducing both MR signal increase and fluorescence recovery due to free Mn2+ ions released from Au clusters. This Au-MnO2 nanocomposite-based magnetic/fluorometric bimodal biosensor allows detection of AA in human serum with cross-validation.
Interactions of the UFO-shaped 2D Au-MnO2 nanostructures with different living cells probed by dark-field images and scattering spectra: (A-E) HepG2 cells, (F-J) 3T3 cells, and (K-O) buthionine sulfoximine (a GSH inhibitor) pre-treated HepG2 cells. (D, I, N) Time-dependent λmax of the scattering spectra changes (E, J, O) after entering the cells. (P) Schematic for two different types of transmembrane processes. (Q) TEM image of UFO-shaped 2D Au-MnO2 nanostructures. Images reproduced with permission from [163], copyright 2019 John Wiley and Sons.
Other Au NPs-based nanocomposites
In addition to the above mentioned hybrid nanostructures, some other Au-based nanocomposites including Au-ZnO, Au-TiO2, and Au-reduced graphene oxide (rGO) have also been developed and utilized as new type of theranostic platforms for biomedical application [165-169]. Metal oxide NPs such as ZnO and TiO2 can absorb photons and create electron-hole pairs, generating ROS to inhibit microbial or cancer growth. However, they only absorb in the UV region, and their capability of ROS generation is limited by fast electron-hole recombination. By integrating ZnO or TiO2 with Au NPs, their optical absorption can be enhanced due to LSPR effect, the spectral window is extended to the visible, and the photo generated charge carrier recombination is greatly suppressed, leading to enhanced photocatalytic and PDT activity. For example, Yin et al. synthesized ZnO/Au hybrid nanostructures using a photo-reduction method [165]. It was found that even Au NPs of sizes less than 3 nm deposited on ZnO NPs can greatly enhance the photo-induced charge carriers in ZnO NPs and thus promoting their ROS generation. Their result demonstrated that constructing hybrid nanostructures with Au is an efficient strategy to improve the photodynamic therapeutic effect of metal oxide. Au-TiO2 NPs is another Au-based hybrid nanostructure with certain physicochemical properties outperforming their respective building blocks. For example, Yin et al. designed Au-TiO2 nanostructures and explored their ROS generation capacity under ultrasound stimulation [166]. Their results revealed that the hybrid structures exhibited higher ROS generation efficiency and more significant tumor suppression effect than their counterparts without Au growth, demonstrating the potential of using Au-TiO2 nanocomposite as sonosensitizer for cancer therapy. Another interesting Au NPs based-hybrid nanostructure is Au-rGO, as demonstrated by Lim et al., where rGO was coated over Au nanorod [167, 168]. Due to high thermal conductivity of rGO and LSPR characteristic of Au nanorods, the prepared Au-rGO hybrid nanostructures exhibited amplified photothermal effect and PA signal intensity, compared to pure Au or graphene oxide/reduced graphene oxide. Moreover, Au can also be integrated with silica layer to form Au@silica nanostructure [169], thus combining the high drug loading capacity of silica with strong photothermal response of Au nanostructures, leading to better cancer cell killing outcome due to the synergistic effect of photothermia and NIR-induced drug release.
Limitations and challenges
As reviewed briefly, many Au-based nanohybrids with enhanced physicochemical properties and bioactivities have been developed to date, which possess the potential to significantly improve cancer treatment outcomes. However, many challenges need to be resolved before they can be successfully translated to clinical usages.
Synthetic challenges
Many synthetic issues remain to be addressed before we can explore the nanohybrids unique properties for nanomedicine. Although a rich library of noble metal Au-based nanocomposites is now available, their syntheses are generally complex with many reaction variables to tune. One critical question is how to establish a facile and general synthetic method that can build up the nanohybrids with the right functional building blocks of proper size, interface, and geometry [170]. Currently, the typical seeded growth route relies on depositing the second component on the seed NP nucleated in situ or synthesized in advance, which can be severely limited by the interfacial energy or lattice matching requirements of the different crystalline domains. Moreover, the growth kinetics can be influenced by complicate synthetic conditions such as reaction temperature, concentration ratio of growth material to seeds NPs, and surface property of the seeds. Non-optimized procedures may lead to low yield of nanohybrids at the end of long tedious procedures. Therefore, developing general synthetic route and establishing standardized protocols to reliably prepare high-quality noble metal-based nanocomposites with controllable morphologies is highly desirable for their extensive biomedical applications.
Biosafety
For clinical applications, the critical pharmacological behaviors such as biodistribution and biosafety of inorganic nanomaterials remain an under-explored territory. The physicochemical attributes such as NP size, shape, and surface coating are known to affect their cellular uptake, biodistribution, and nanotoxicity. This calls for systematic investigation on the in vivo behaviors of designed nanocomposites. The choice of chemical composition and surface coating is clearly critical for the nanocomposites biocompatibility. In terms of composition, Au NPs are generally considered to be bioinert, while copper chalcogenides and metal oxides may be etched or biodegraded in the body fluid, releasing metal ions and introducing potential toxicity to cells and organs. On the other hand, nanotoxicity is also strongly influenced by NP surface modifications. Surface coating can induce cytotoxicity effect directly or indirectly by influencing the formation of protein corona, and the subsequent cellular internalization and final fate of the NPs. As many of the NP physicochemical properties are highly interconnected, it is challenging to evaluate the cytotoxic effect originating from one single attribute of the NPs. Moreover, issues on the long-term metabolism of inorganic nanomaterials such as decomposition, degradation, and clearance of the nanocomposites from the body need to be addressed before they can be applied for clinical usage [171-177]. Although many cytotoxicity studies on Au NPs have suggested that they possess good biological safety within several weeks, a great risk of the bioinert NPs is that they may stay in the body and induce chronic toxicity over extended time. Therefore, a balanced stability, slow degradation, and fast clearance should be considered for nanohybrids design with proper choice of chemical composition and surface coating. Finally, the in vitro/in vivo models employed in the biosafety evaluation can also influence the behavior and fate of the hybrid NPs, which may render conflicting results. To obtain accurate and consistent nanotoxicity evaluation, establishing standardized and reliable protocols to systematically investigate the impact of pharmacological parameters of the NPs is fundamentally important for the biosafety study of the hybrid NPs.
Conclusions and perspectives
Nanohybrids composed of noble metal Au and copper chalcogenides or magnetic metal oxides have emerged as a unique class of material due to their interesting plasmon-magnetic properties, and the combined diagnostic and therapeutic functional units in one single entity. In this short review, we have summarized some recent developments in building up Au-based inorganic nanohybrids with controlled composition and structure, and highlighted progresses made in their theranostics applications.
Despite substantial progresses that have been made in the field of Au-based hybrid nanomedicine, this field is still at a rather preliminary stage from the standpoint of practical medical applications, especially in terms of biosafety that we have pointed out in the previous section. To address these important questions, several issues need to be addressed. First of all, more comprehensive studies need to be focused on the nanomaterial-biological system interactions, in order to better understand the critical factors determining the biosafety of nanocomposites, which will then be used for better nanomedicine design. Although plenty nanotoxicity studies have already been carried out on Au [36, 178-181], whether the attachment of a second component would affect its cellular behavior and final fate within organisms is still uncertain. It is believed that many physicochemical parameters of nanomaterials such as size, shape, charge, and surface modification can greatly influence the biocompatibility of nanocomposites. Future toxicity investigations on noble metal Au-based nanocomposites should consider all of these complex factors and explore the underlying molecular mechanisms of various factors on gene expression, signaling pathways, and downstream cell metabolism. In addition, when interpreting the interaction between nanocomposites and organism, it is necessary to note that the organisms may behave well and show normal physiological functions for a short period [182]. However, the organisms may suffer subtle but irreversible changes in their genetics after continuous exposure. Therefore, additional long-term toxicity evaluation is needed in the corresponding animal experiments. Moreover, excellent bioavailability and targeting ability is of great importance for highly efficient biomedical nanotechnologies. The hybrid nanocomposites must avoid rapid clearance during blood circulation and increase their accumulation dosage at the desirable target site. Recently, the cell membrane-cloaking strategy by mimicking nanoparticles with erythrocyte or host cancer cell membrane envelopes has shown great potency in increasing circulation time by inhibiting macrophage recognition and improving targeting ability via homotypic binding [183-187]. However, this technique still faces some inherent challenges. For example, the detailed biomolecular mechanism of the homotypic binding derived from cell membrane is yet unclear. Identification of the specific ligands involved in the host membrane recognition would benefit future development of nanomaterial-based biomimetic nanotechnology. Furthermore, it is very difficult for the nanohybrids to go deep into solid tumors, which severely limits their efficacy as drug carrier and imaging platform. Knowledge on the NPs' pathway into tumors would be useful in helping design nanohybrids structures with improved tumor penetration depth. Despite the discovery of endothelial gaps in tumor vasculatures using developed animal models, nanomedicine design utilizing the enhanced permeability and retention (EPR) effect for human tumor treatment has been controversial. Only a few anticancer nanomedicines have received approval for clinical application based on EPR effect. Recently, new evidence has emerged, suggesting NPs may enter tumors via an active process through endothelial transcytosis [188]. These observations may establish new paradigms and enable novel strategies to help expedite the clinical translation of nanomedicine.
Au-based multifunctional nanocomposites have shown their promises in both early diagnostic and theranostic applications. Of note, their manifested multifunctionality due to the synergistic effect between different components would enable safer and more effective theranostic treatment. Undoubtedly, with continuing endeavor in the design and development of new multifunctional nanohybrids, it is our firm belief that they hold great diagnostic and therapeutic potential in broad biomedical applications, and are likely to find real significance in the new era of personalized precision nanomedicine.
Acknowledgements
This work was funded by Six Talent Peaks Project in Jiangsu Province (SWYY-243) and National Natural Science Foundation of China (No. 21873113). X.G. Ding also acknowledges the start-up fund from Nanjing University of Posts and Telecommunications.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Naghavi M, Abajobir A, Abbafati C, Abbas K, Abd-Allah F, Abera S. GBD 2016 causes of death collaborators. global, regional, and national age-sex specific mortality for 264 causes of death, 1980-2016: a systematic analysis for the global burden of disease study 2016. Lancet. 2017;390:1151-1210
2. Bray F, Ferlay J, Soerjomataram I, Siegel R L, Torre L A, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424
3. Torre LA, Trabert B, DeSantis CE, Miller KD, Samimi G, Runowicz CD. et al. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018;68:284-296
4. Zhang J, Jiang C, Longo JPF, Azevedo RB, Zhang H, Muehlmann LA. An updated overview on the development of new photosensitizers for anticancer photodynamic therapy. Acta Pharm Sin B. 2018;8:137-146
5. Zhang Z, Wang J, Chen C. Gold nanorods based platforms for light-mediated theranostics. Theranostics. 2013;3:223-238
6. Chen M, Tang S, Guo Z, Wang X, Mo S, Huang X. et al. Core-Shell Pd@Au Nanoplates as theranostic agents for in-vivo photoacoustic imaging, CT imaging, and photothermal therapy. Adv Mater. 2014;26:8210-8216
7. Zhang P, Hu C, Ran W, Meng J, Yin Q, Li Y. Recent progress in light-triggered nanotheranostics for cancer treatment. Theranostics. 2016;6:948-968
8. Liu Y, Yang X, Huang Z, Huang P, Zhang Y, Deng L. et al. Magneto-plasmonic janus vesicles for magnetic field-enhanced photoacoustic and magnetic resonance imaging of tumors. Angew Chem Int Ed. 2016;55:15297-15300
9. Jin J, Ovais M, Chen C. Stimulus-responsive gold nanotheranostic platforms for targeting the tumor microenvironment. Nano Today. 2018;22:83-99
10. Sun W, Luo L, Feng Y, Cai Y, Zhuang Y, Xie RJ. et al. Aggregation-induced emission gold clustoluminogens for enhanced low-dose X-ray-induced photodynamic therapy. Angew Chem Int Ed. 2020;59:9914-9921
11. Yang K, Liu Y, Wang Y, Ren Q, Guo H, Matson JB. et al. Enzyme-induced in vivo assembly of gold nanoparticles for imaging-guided synergistic chemo-photothermal therapy of tumor. Biomaterials. 2019;223:119460
12. Zhang Z, Yao Y, Yuan Q, Lu C, Zhang X, Yuan J. et al. Gold clusters prevent breast cancer bone metastasis by suppressing tumor-induced osteoclastogenesis. Theranostics. 2020;10:4042-4055
13. Ding Y, Sun Z, Tong Z, Zhang S, Min J, Xu Q. et al. Tumor microenvironment-responsive multifunctional peptide coated ultrasmall gold nanoparticles and their application in cancer radiotherapy. Theranostics. 2020;10:5195-5208
14. Ge X, Fu Q, Su L, Li Z, Zhang W, Chen T. et al. Light-activated gold nanorod vesicles with NIR-II fluorescence and photoacoustic imaging performances for cancer theranostics. Theranostics. 2020;10:4809-4821
15. Hu M, Chen J, Li ZY, Au L, Hartland GV, Li X. et al. Gold nanostructures: engineering their plasmonic properties for biomedical applications. Chem Soc Rev. 2006;35:1084-1094
16. Maier SA, Plasmonics. fundamentals and applications. Springer Science & Business Media. 2007
17. Anker JN, Hall WP, Lyandres O, Shah NC, Zhao J, Van Duyne RP, Biosensing with plasmonic nanosensors. In nanoscience and technology: a collection of reviews from nature journals, World Scientific. 2010; p:308-319.
18. Giannini V, Fernández-Domínguez AI, Heck SC, Maier SA. Plasmonic nanoantennas: fundamentals and their use in controlling the radiative properties of nanoemitters. Chem Rev. 2011;111:3888-3912
19. Qiu K, Du Y, Liu J, Guan JL, Chao H, Diao J. Super-resolution observation of lysosomal dynamics with fluorescent gold nanoparticles. Theranostics. 2020;10:6072-6081
20. Ye H, Liu Y, Zhan L, Liu Y, Qin Z. Signal amplification and quantification on lateral flow assays by laser excitation of plasmonic nanomaterials. Theranostics. 2020;10:4359-4373
21. Zhang A, Pan S, Zhang Y, Chang J, Cheng J, Huang Z. et al. Carbon-gold hybrid nanoprobes for real-time imaging, photothermal/photodynamic and nanozyme oxidative therapy. Theranostics. 2019;9:3443-3458
22. Boisselier E, Astruc D. Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chem Soc Rev. 2009;38:1759-1782
23. Zhang XF, Liu ZG, Shen W, Gurunathan S. Silver nanoparticles: synthesis, characterization, properties, applications, and therapeutic approaches. Int J Mol Sci. 2016;17:1534
24. Carabineiro SAC. Applications of gold nanoparticles in nanomedicine: Recent advances in vaccines. Molecules. 2017;22:857
25. Skrabalak SE, Au L, Li X, Xia Y. Facile synthesis of Ag nanocubes and Au nanocages. Nat Protoc. 2007;2:2182-2190
26. Millstone JE, Hurst SJ, Métraux GS, Cutler JI, Mirkin CA. Colloidal gold and silver triangular nanoprisms. Small. 2009;5:646-664
27. Xia Y, Li W, Cobley CM, Chen J, Xia X, Zhang Q. et al. Gold nanocages: from synthesis to theranostic applications. Acc Chem Res. 2011;44:914-924
28. Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Rivera B, Price R. et al. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. P Natl Acad Sci USA. 2003;100:13549-13554
29. Loo C, Lowery A, Halas N, West J, Drezek R. Immunotargeted nanoshells for integrated cancer imaging and therapy. Nano Lett. 2005;5:709-711
30. Kinnear C, Moore TL, Rodriguez-Lorenzo L, Rothen-Rutishauser B, Petri-Fink A. Form follows function: nanoparticle shape and its implications for nanomedicine. Chem Rev. 2017;117:11476-11521
31. Jain PK, Huang X, El-Sayed IH, El-Sayed MA. Noble metals on the nanoscale: optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc Chem Res. 2008;41:1578-1586
32. Jain PK, El-Sayed MA. Plasmonic coupling in noble metal nanostructures. Chem Phys Lett. 2010;487:153-164
33. Liu X, Zhang X, Zhu M, Lin G, Liu J, Zhou Z. et al. PEGylated Au@Pt nanodendrites as novel theranostic agents for computed tomography imaging and photothermal/radiation synergistic therapy. ACS Appl Mater Interfaces. 2017;9:279-285
34. Deng Y, Tian X, Lu S, Xie M, Hu H, Zhang R. et al. Fabrication of multifoliate PtRu bimetallic nanocomplexes for computed tomography imaging and enhanced synergistic thermoradiotherapy. ACS Appl Mater Interfaces. 2018;10:31106-31113
35. Soliman MG, Pelaz B, Parak WJ, Del Pino P. Phase transfer and polymer coating methods toward improving the stability of metallic nanoparticles for biological applications. Chem Mater. 2015;27:990-997
36. Murphy CJ, Gole AM, Stone JW, Sisco PN, Alkilany AM, Goldsmith EC. et al. Gold nanoparticles in biology: beyond toxicity to cellular imaging. Acc Chem Res. 2008;41:1721-1730
37. Dou Y, Guo Y, Li X, Li X, Wang S, Wang L. et al. Size-tuning ionization to optimize gold nanoparticles for simultaneous enhanced CT imaging and radiotherapy. ACS Nano. 2016;10:2536-2548
38. Chhour P, Kim J, Benardo B, Tovar A, Mian S, Litt HI. et al. Effect of gold nanoparticle size and coating on labeling monocytes for CT tracking. Bioconj Chem. 2017;28:260-269
39. Ashton JR, Castle KD, Qi Y, Kirsch DG, West JL, Badea CT. Dual-energy CT imaging of tumor liposome delivery after gold nanoparticle-augmented radiation therapy. Theranostics. 2018;8:1782-1797
40. Yang X, Skrabalak SE, Li ZY, Xia Y, Wang LV. Photoacoustic tomography of a rat cerebral cortex in vivo with Au nanocages as an optical contrast agent. Nano Lett. 2007;7:3798-3802
41. Fu Q, Zhu R, Song J, Yang H, Chen X. Photoacoustic imaging: contrast agents and their biomedical applications. Adv Mater. 2019;31:1805875
42. Skrabalak SE, Au L, Lu X, Li X, Xia Y. Gold nanocages for cancer detection and treatment. Nanomedicine. 2007;2:657-668
43. Sun T, Zhang YS, Pang B, Hyun DC, Yang M, Xia Y. Engineered nanoparticles for drug delivery in cancer therapy. Angew Chem Int Ed. 2014;53:12320-12364
44. Yang M, Wang W, Qiu J, Bai MY, Xia Y. Direct visualization and semi-quantitative analysis of payload loading in the case of gold nanocages. Angew Chem Int Ed. 2019;58:17671-17674
45. Song G, Cheng L, Chao Y, Yang K, Liu Z. Emerging nanotechnology and advanced materials for cancer radiation therapy. Adv Mater. 2017;29:1700996
46. Espinosa A, Curcio A, Cabana S, Radtke G, Bugnet M, Kolosnjaj-Tabi J. et al. Intracellular biodegradation of Ag nanoparticles, storage in ferritin, and protection by a Au shell for enhanced photothermal therapy. ACS Nano. 2018;12:6523-6535
47. Xi Z, Ye H, Xia X. Engineered noble-metal nanostructures for in vitro diagnostics. Chem Mater. 2018;30:8391-8414
48. Xi Z, Cheng X, Gao Z, Wang M, Cai T, Muzzio M. et al. Strain effect in palladium nanostructures as nanozymes. Nano Lett. 2019;20:272-277
49. Wang X, Qin L, Zhou M, Lou Z, Wei H. Nanozyme sensor arrays for detecting versatile analytes from small molecules to proteins and cells. Anal Chem. 2018;90:11696-11702
50. Hu Y, Cheng H, Zhao X, Wu J, Muhammad F, Lin S. et al. Surface-enhanced Raman scattering active gold nanoparticles with enzyme-mimicking activities for measuring glucose and lactate in living tissues. ACS Nano. 2017;11:5558-5566
51. Shaviv E, Schubert O, Alves-Santos M, Goldoni G, Di Felice R, Vallée F. et al. Absorption properties of metal-semiconductor hybrid nanoparticles. ACS Nano. 2011;5:4712-4719
52. Khon E, Mereshchenko A, Tarnovsky AN, Acharya K, Klinkova A, Hewa-Kasakarage NN. et al. Suppression of the plasmon resonance in Au/CdS colloidal nanocomposites. Nano Lett. 2011;11:1792-9
53. Zhang Z, Yates Jr JT. Band bending in semiconductors: chemical and physical consequences at surfaces and interfaces. Chem Rev. 2012;112:5520-5551
54. Mokari T, Sztrum CG, Salant A, Rabani E, Banin U. Formation of asymmetric one-sided metal-tipped semiconductor nanocrystal dots and rods. Nat Mater. 2005;4:855-863
55. Li Y, Wen T, Zhao R, Liu X, Ji T, Wang H. et al. Localized electric field of plasmonic nanoplatform enhanced photodynamic tumor Therapy. ACS Nano. 2014;8:11529-11542
56. Song J, Yang X, Jacobson O, Lin L, Huang P, Niu G. et al. Sequential drug release and enhanced photothermal and photoacoustic effect of hybrid reduced graphene oxide-loaded ultrasmall gold nanorod vesicles for cancer therapy. ACS Nano. 2015;9:9199-9209
57. Ji M, Xu M, Zhang W, Yang Z, Huang L, Liu J. et al. Structurally well-defined Au@Cu2-xS core-shell nanocrystals for improved cancer treatment based on enhanced photothermal efficiency. Adv Mater. 2016;28:3094-3101
58. Park S, Lee WJ, Park S, Choi D, Kim S, Park N. Reversibly pH-responsive gold nanoparticles and their applications for photothermal cancer therapy. Sci Rep. 2019;9:20180
59. Rossi A, Donati S, Fontana L, Porcaro F, Battocchio C, Proietti E. et al. Negatively charged gold nanoparticles as a dexamethasone carrier: stability in biological media and bioactivity assessment in vitro. RSC Adv. 2016;6:99016-99022
60. Yang W, Liang H, Ma S, Wang D, Huang J. Gold nanoparticle based photothermal therapy: Development and application for effective cancer treatment. Sustainable Mater Technol. 2019;22:e00109
61. Porcaro F, Battocchio C, Antoccia A, Fratoddi I, Venditti I, Fracassi A. et al. Synthesis of functionalized gold nanoparticles capped with 3-mercapto-1-propansulfonate and 1-thioglucose mixed thiols and "in vitro" bioresponse. Colloids Surf B. 2016;142:408-416
62. Grabowska-Jadach I, Kalinowska D, Drozd M, Pietrzak M. Synthesis, characterization and application of plasmonic hollow gold nanoshells in a photothermal therapy-new particles for theranostics. Biomed Pharmacother. 2019;111:1147-1155
63. Fratoddi I, Venditti I, Battocchio C, Carlini L, Amatori S, Porchia M. et al. Highly hydrophilic gold nanoparticles as carrier for anticancer copper(I) complexes: loading and release studies for biomedical applications. Nanomaterials. 2019;9:772
64. Khlebtsov N, Bogatyrev V, Dykman L, Khlebtsov B, Staroverov S, Shirokov A. et al. Analytical and theranostic applications of gold nanoparticles and multifunctional nanocomposites. Theranostics. 2013;3:167-80
65. Venditti I. Engineered gold-based nanomaterials: morphologies and functionalities in biomedical applications. a mini review. Bioengineering. 2019;6:53
66. Maccora D, Dini V, Battocchio C, Fratoddi I, Cartoni A, Rotili D. et al. Gold nanoparticles and nanorods in nuclear nedicine: a mini review. Applied Sciences. 2019;9:3232
67. Ma K, Li Y, Wang Z, Chen Y, Zhang X, Chen C. et al. Core-shell gold nanorod@layered double hydroxide nanomaterial with highly efficient photothermal conversion and its application in antibacterial and tumor therapy. ACS Appl Mater Interfaces. 2019;11:29630-29640
68. Chen W, Zhang S, Yu Y, Zhang H, He Q. Structural-engineering rationales of gold nanoparticles for cancer theranostics. Adv Mater. 2016;28:8567-8585
69. Ju Y, Zhang H, Yu J, Tong S, Tian N, Wang Z. et al. Monodisperse Au-Fe2C Janus nanoparticles: an attractive multifunctional material for triple-modal imaging-guided tumor photothermal therapy. ACS Nano. 2017;11:9239-9248
70. Reguera J, de Aberasturi DJ, Henriksen-Lacey M, Langer J, Espinosa A, Szczupak B. et al. Janus plasmonic-magnetic gold-iron oxide nanoparticles as contrast agents for multimodal imaging. Nanoscale. 2017;9:9467-9480
71. Kim D, Shin K, Kwon S G, Hyeon T. Synthesis and biomedical applications of multifunctional nanoparticles. Adv Mater. 2018;30:1802309
72. Zeng J, Gong M, Wang D, Li M, Xu W, Li Z. et al. Direct synthesis of water-dispersible magnetic/plasmonic heteronanostructures for multimodality biomedical imaging. Nano Lett. 2019;19:3011-3018
73. Lv Q, Min H, Duan DB, Fang W, Pan GM, Shen AG. et al. Total aqueous synthesis of Au@Cu2-xS core-shell nanoparticles for in vitro and in vivo SERS/PA imaging-guided photothermal cancer therapy. Adv Healthc Mater. 2019;8:1801257
74. Jauffred L, Samadi A, Klingberg H, Bendix PM, Oddershede LB. Plasmonic heating of nanostructures. Chem Rev. 2019;119:8087-8130
75. Lee SE, Lee LP. Biomolecular plasmonics for quantitative biology and nanomedicine. Curr Opin Biotechnol. 2010;21:489-497
76. Lim WQ, Gao Z. Plasmonic nanoparticles in biomedicine. Nano Today. 2016;11:168-188
77. Ding X, Liow CH, Zhang M, Huang R, Li C, Shen H. et al. Surface plasmon resonance enhanced light absorption and photothermal therapy in the second near-infrared window. J Am Chem Soc. 2014;136:15684-15693
78. Liu X, Lee C, Law WC, Zhu D, Liu M, Jeon M. et al. Au-Cu2-xSe heterodimer nanoparticles with broad localized surface plasmon resonance as contrast agents for deep tissue imaging. Nano Lett. 2013;13:4333-4339
79. Song J, Lin L, Yang Z, Zhu R, Zhou Z, Li ZW. et al. Self-assembled responsive bilayered vesicles with adjustable oxidative stress for enhanced cancer imaging and therapy. J Am Chem Soc. 2019;141:8158-8170
80. Ding X, Zhao H, Li C, Wang Q, Jiang J. All-in-one theranostic nanoplatform with controlled drug release and activated MRI tracking functions for synergistic NIR-II hyperthermia-chemotherapy of tumors. Nano Res. 2019;12:2971-2981
81. Tao C, An L, Lin J, Tian Q, Yang S. Surface plasmon resonance-enhanced photoacoustic imaging and photothermal therapy of endogenous H2S-triggered Au@Cu2O. Small. 2019;15:1903473
82. Zhao Y, Pan H, Lou Y, Qiu X, Zhu J, Burda C. Plasmonic Cu2-xS nanocrystals: optical and structural properties of copper-deficient copper (I) sulfides. J Am Chem Soc. 2009;131:4253-4261
83. Luther JM, Jain PK, Ewers T, Alivisatos AP. Localized surface plasmon resonances arising from free carriers in doped quantum dots. Nat Mater. 2011;10:361-366
84. Comin A, Manna L. New materials for tunable plasmonic colloidal nanocrystals. Chem Soc Rev. 2014;43:3957-3975
85. Buonsanti R, Milliron DJ. Chemistry of doped colloidal nanocrystals. Chem Mater. 2013;25:1305-1317
86. Agrawal A, Cho SH, Zandi O, Ghosh S, Johns RW, Milliron DJ. Localized surface plasmon resonance in semiconductor nanocrystals. Chem Rev. 2018;118:3121-3207
87. Liu Z, Liu X, Du Y, Ren J, Qu X. Using plasmonic copper sulfide nanocrystals as smart light-driven sterilants. ACS Nano. 2015;9:10335-10346
88. Yang W, Guo W, Le W, Lv G, Zhang F, Shi L. et al. Albumin-bioinspired Gd: CuS nanotheranostic agent for in vivo photoacoustic/magnetic resonance imaging-guided tumor-targeted photothermal therapy. ACS Nano. 2016;10:10245-10257
89. Kumar A, Kim S, Nam JM. Plasmonically engineered nanoprobes for biomedical applications. J Am Chem Soc. 2016;138:14509-14525
90. Kriegel I, Scotognella F, Manna L. Plasmonic doped semiconductor nanocrystals: Properties, fabrication, applications and perspectives. Phys Rep. 2017;674:1-52
91. Shan B, Zhao Y, Li Y, Wang H, Chen R, Li M. High-quality dual-plasmonic Au@Cu2-xSe nanocrescents with precise Cu2-xSe domain size control and tunable optical properties in the second near-infrared biowindow. Chem Mater. 2019;31:9875-9886
92. Ding X, Fu D, Kuang Y, Zou Y, Yang X, Feng L. et al. Seeded growth of Cu2-xSe nanocrystals and their size-dependent phototherapeutic effect. ACS Appl Nano Mater. 2018;1:3303-3311
93. Ou W, Zou Y, Wang K, Gong W, Pei R, Chen L. et al. Active manipulation of NIR plasmonics: the case of Cu2-xSe through electrochemistry. J Phys Chem Lett. 2018;9:274-280
94. Sun X, Zou Y, Jiang J. Surface plasmon resonances enhanced click chemistry through synergistic photothermal and hot electron effects. Chem Commun. 2019;55:4813-4816
95. Ji M, Xu M, Zhang W, Yang Z, Huang L, Liu J. et al. Structurally well-defined Au@Cu2-xS core-shell nanocrystals for improved cancer treatment based on enhanced photothermal efficiency. Adv Mater. 2016;28:3094-3101
96. Deng X, Li K, Cai X, Liu B, Wei Y, Deng K. et al. A Hollow-structured CuS@Cu2S@Au nanohybrid: synergistically enhanced photothermal efficiency and photoswitchable targeting effect for cancer theranostics. Adv Mater. 2017;29:1701266
97. Bashkatov A, Genina E, Kochubey V, Tuchin V. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J Phys D: Appl Phys. 2005;38:2543
98. Hong G, Robinson JT, Zhang Y, Diao S, Antaris AL, Wang Q. et al. In vivo fluorescence imaging with Ag2S quantum dots in the second near-infrared region. Angew Chem Int Ed. 2012;51:9818-9821
99. Smith AM, Mancini MC, Nie S. Bioimaging: second window for in vivo imaging. Nat Nanotechnol. 2009;4:710-711
100. Welsher K, Liu Z, Sherlock SP, Robinson JT, Chen Z, Daranciang D. et al. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat Nanotechnol. 2009;4:773-780
101. Huang J, Xie C, Zhang X, Jiang Y, Li J, Fan Q. et al. Renal-clearable molecular semiconductor for second near-infrared fluorescence imaging of kidney dysfunction. Angew Chem Int Ed. 2019;58:15120-15127
102. Zou Y, Sun C, Gong W, Yang X, Huang X, Yang T. et al. Morphology-controlled synthesis of hybrid nanocrystals via a selenium-mediated strategy with ligand shielding effect: the case of dual plasmonic Au-Cu2-xSe. ACS Nano. 2017;11:3776-3785
103. Zhu H, Wang Y, Chen C, Ma M, Zeng J, Li S. et al. Monodisperse dual plasmonic Au@Cu2-xE (E= S, Se) core@ shell supraparticles: aqueous fabrication, multimodal imaging, and tumor therapy at in vivo level. ACS Nano. 2017;11:8273-8281
104. Li W, Chen X. Gold nanoparticles for photoacoustic imaging. Nanomedicine. 2015;10:299-320
105. Weber J, Beard PC, Bohndiek SE. Contrast agents for molecular photoacoustic imaging. Nat Methods. 2016;13:639-650
106. Wang LV, Hu S. Photoacoustic tomography: in vivo imaging from organelles to organs. Science. 2012;335:1458-1462
107. Nagarathinam M, Chen J, Vittal JJ. From self-assembled Cu (II) coordination polymer to shape-controlled CuS nanocrystals. Cryst Growth Des. 2009;9:2457-2463
108. Gao D, Sheng Z, Liu Y, Hu D, Zhang J, Zhang X. et al. Protein-modified CuS nanotriangles: a potential multimodal nanoplatform for in vivo tumor photoacoustic/magnetic resonance dual-modal imaging. Adv Healthc Mater. 2017;6:1601094
109. Lv Q, Gao MY, Cheng ZH, Chen Q, Shen AG, Hu JM. Rational synthesis of hollow cubic CuS@Spiky Au core-shell nanoparticles for enhanced photothermal and SERS effects. Chem Commun. 2018;54:13399-13402
110. Chang Y, Cheng Y, Feng Y, Jian H, Wang L, Ma X. et al. Resonance energy transfer-promoted photothermal and photodynamic performance of gold-copper sulfide yolk-shell nanoparticles for chemophototherapy of cancer. Nano Lett. 2018;18:886-897
111. Cao Y, Li S, Chen C, Wang D, Wu T, Dong H. et al. Rattle-type Au@Cu2-xS hollow mesoporous nanocrystals with enhanced photothermal efficiency for intracellular oncogenic microRNA detection and chemo-photothermal therapy. Biomaterials. 2018;158:23-33
112. LaConte L, Nitin N, Bao G. Magnetic nanoparticle probes. Mater Today. 2005;8:32-38
113. Jiang J, Gu H, Shao H, Devlin E, Papaefthymiou G C, Ying JY. Bifunctional Fe3O4-Ag heterodimer nanoparticles for two-photon fluorescence imaging and magnetic manipulation. Adv Mater. 2008;20:4403-4407
114. Shao H, Min C, Issadore D, Liong M, Yoon TJ, Weissleder R. et al. Magnetic nanoparticles and microNMR for diagnostic applications. Theranostics. 2012;2:55-65
115. Hervault A, Thanh NTK. Magnetic nanoparticle-based therapeutic agents for thermo-chemotherapy treatment of cancer. Nanoscale. 2014;6:11553-11573
116. Shin TH, Choi Y, Kim S, Cheon J. Recent advances in magnetic nanoparticle-based multi-modal imaging. Chem Soc Rev. 2015;44:4501-4516
117. Chen Y, Ding X, Zhang Y, Natalia A, Sun X, Wang Z. et al. Design and synthesis of magnetic nanoparticles for biomedical diagnostics. Quant Imag Med Surg. 2018;8:957-970
118. Wang X, Zhang C, Du J, Dong X, Jian S, Yan L. et al. Enhanced generation of non-oxygen dependent free radicals by schottky-type heterostructures of Au-Bi2S3 nanoparticles via X-ray-induced catalytic reaction for radiosensitization. ACS Nano. 2019;13:5947-5958
119. Yang Z, Ding X, Jiang J. Facile synthesis of magnetic-plasmonic nanocomposites as T1 MRI contrast enhancing and photothermal therapeutic agents. Nano Res. 2016;9:787-799
120. Deatsch AE, Evans BA. Heating efficiency in magnetic nanoparticle hyperthermia. J Magn Magn Mater. 2014;354:163-172
121. Mohammad F, Balaji G, Weber A, Uppu RM, Kumar CS. Influence of gold nanoshell on hyperthermia of superparamagnetic iron oxide nanoparticles. J Phys Chem C. 2010;114:19194-19201
122. Han Y, Lei SL, Lu JH, He Y, Chen ZW, Ren L. et al. Potential use of SERS-assisted theranostic strategy based on Fe3O4/Au cluster/shell nanocomposites for bio-detection, MRI, and magnetic hyperthermia. Mat Sci Eng: C. 2016;64:199-207
123. Li Z, Aranda-Ramos A, Güell-Grau P, Tajada JL, Pou-Macayo L, Piedrafita SL. et al. Magnetically amplified photothermal therapies and multimodal imaging with magneto-plasmonic nanodomes. Appl Mater Today. 2018;12:430-440
124. Espinosa A, Bugnet M, Radtke G, Neveu S, Botton GA, Wilhelm C. et al. Can magneto-plasmonic nanohybrids efficiently combine photothermia with magnetic hyperthermia? Nanoscale. 2015;7:18872-18877
125. Tang L, Yang X, Dobrucki LW, Chaudhury I, Yin Q, Yao C. et al. Aptamer-functionalized, ultra-small, monodisperse silica nanoconjugates for targeted dual-modal imaging of lymph nodes with metastatic tumors. Angew Chem Int Ed. 2012;51:12721-12726
126. Wong RM, Gilbert DA, Liu K, Louie A Y. Rapid size-controlled synthesis of dextran-coated, 64Cu-doped iron oxide nanoparticles. ACS Nano. 2012;6:3461-3467
127. Chen F, Ellison PA, Lewis CM, Hong H, Zhang Y, Shi S. et al. Chelator-free synthesis of a dual-modality PET/MRI agent. Angew Chem Int Ed. 2013;52:13319-13323
128. Chen Q, Li K, Wen S, Liu H, Peng C, Cai H. et al. Targeted CT/MR dual mode imaging of tumors using multifunctional dendrimer-entrapped gold nanoparticles. Biomaterials. 2013;34:5200-5209
129. Hu DH, Sheng ZH, Zhang PF, Yang DZ, Liu SH, Gong P. et al. Hybrid gold-gadolinium nanoclusters for tumor-targeted NIRF/CT/MRI triple-modal imaging in vivo. Nanoscale. 2013;5:1624-1628
130. Wei Q, Chen Y, Ma X, Ji J, Qiao Y, Zhou B. et al. High-efficient clearable nanoparticles for multi-modal imaging and image-guided cancer therapy. Adv Funct Mater. 2018;28:1704634
131. Chen Q, Wen J, Li H, Xu Y, Liu F, Sun S. Recent advances in different modal imaging-guided photothermal therapy. Biomaterials. 2016;106:144-166
132. Liu H, Lin W, He L, Chen T. Radiosensitive core/satellite ternary heteronanostructure for multimodal imaging-guided synergistic cancer radiotherapy. Biomaterials. 2020;226:119545
133. Narayanan S, Sathy BN, Mony U, Koyakutty M, Nair SV, Menon D. Biocompatible magnetite/gold nanohybrid contrast agents via green chemistry for MRI and CT bioimaging. ACS Appl Mater Interfaces. 2012;4:251-260
134. Yang M, Cheng K, Qi S, Liu H, Jiang Y, Jiang H. et al. Affibody modified and radiolabeled gold-iron oxide hetero-nanostructures for tumor PET, optical and MR imaging. Biomaterials. 2013;34:2796-2806
135. Cai H, Li K, Li J, Wen S, Chen Q, Shen M. et al. Dendrimer-assisted formation of Fe3O4/Au nanocomposite particles for targeted dual mode CT/MR imaging of tumors. Small. 2015;11:4584-4593
136. Zhu J, Lu Y, Li Y, Jiang J, Cheng L, Liu Z. et al. Synthesis of Au-Fe3O4 heterostructured nanoparticles for in vivo computed tomography and magnetic resonance dual model imaging. Nanoscale. 2014;6:199-202
137. Li J, Zheng L, Cai H, Sun W, Shen M, Zhang G. et al. Facile one-pot synthesis of Fe3O4@Au composite nanoparticles for dual-mode MR/CT imaging applications. ACS Appl Mater Interfaces. 2013;5:10357-10366
138. He X, Liu F, Liu L, Duan T, Zhang H, Wang Z. Lectin-conjugated Fe2O3@Au core@shell nanoparticles as dual mode contrast agents for in vivo detection of tumor. Mol Pharm. 2014;11:738-745
139. Zhou M, Singhana B, Liu Y, Huang Q, Mitcham T, Wallace MJ. et al. Photoacoustic-and magnetic resonance-guided photothermal therapy and tumor vasculature visualization using theranostic magnetic gold nanoshells. J Biomed Nanotechnol. 2015;11:1442-1450
140. Monaco I, Arena F, Biffi S, Locatelli E, Bortot B, La Cava F. et al. Synthesis of lipophilic core-shell Fe3O4@SiO2@Au nanoparticles and polymeric entrapment into nanomicelles: a novel nanosystem for in vivo active targeting and magnetic resonance-photoacoustic dual imaging. Bioconj Chem. 2017;28:1382-1390
141. Huang J, Guo M, Ke H, Zong C, Ren B, Liu G. et al. Rational design and synthesis of γFe2O3@Au magnetic gold nanoflowers for efficient cancer theranostics. Adv Mater. 2015;27:5049-5056
142. Lin LS, Yang X, Zhou Z, Yang Z, Jacobson O, Liu Y. et al. Yolk-shell nanostructure: an ideal architecture to achieve harmonious integration of magnetic-plasmonic hybrid theranostic platform. Adv Mater. 2017;29:1606681
143. Rosa-Pardo I, Roig-Pons M, Heredia A A, Usagre J, Ribera A, Galian R E. et al. Fe3O4@Au@mSiO2 as an enhancing nanoplatform for Rose Bengal photodynamic activity. Nanoscale. 2017;9:10388-10396
144. Klein S, Smuda M, Harreiss C, Menter C, Distel LVR, Kryschi C. Bifunctional Au-Fe3O4 nanoheterodimers acting as X-ray protector in healthy cells and as X-ray enhancer in tumor cells. ACS Appl Mater Interfaces. 2019;11:39613-39623
145. Gao S, Lin H, Zhang H, Yao H, Chen Y, Shi J. Nanocatalytic Tumor therapy by biomimetic dual inorganic nanozyme-catalyzed cascade reaction. Adv Sci. 2019;6:1801733
146. Wang M, Wang D, Chen Q, Li C, Li Z, Lin J. Recent advances in glucose-oxidase-based nanocomposites for tumor therapy. Small. 2019;15:1903895
147. Schladt TD, Shukoor MI, Schneider K, Tahir MN, Natalio F, Ament I. et al. Au@MnO nanoflowers: hybrid nanocomposites for selective dual functionalization and imaging. Angew Chem Int Ed. 2010;49:3976-3980
148. Song M, Liu T, Shi C, Zhang X, Chen X. Bioconjugated manganese dioxide nanoparticles enhance chemotherapy response by priming tumor-associated macrophages toward M1-like phenotype and attenuating tumor hypoxia. ACS Nano. 2016;10:633-647
149. Lin L S, Song J, Song L, Ke K, Liu Y, Zhou Z. et al. Simultaneous fenton-like ion delivery and glutathione depletion by MnO2-based nanoagent to enhance chemodynamic therapy. Angew Chem Int Ed. 2018;57:4902-4906
150. Liu J, Chen Q, Feng L, Liu Z. Nanomedicine for tumor microenvironment modulation and cancer treatment enhancement. Nano Today. 2018;21:55-73
151. Chen Y, Cong H, Shen Y, Yu B. Biomedical application of manganese dioxide nanomaterials. Nanotechnology. 2020;31:202001
152. Wu J, Williams GR, Niu S, Yang Y, Li Y, Zhang X. et al. Biomineralized bimetallic oxide nanotheranostics for multimodal imaging-guided combination therapy. Theranostics. 2020;10:841-855
153. Fu C, Duan X, Cao M, Jiang S, Ban X, Guo N. et al. Targeted magnetic resonance imaging and modulation of hypoxia with multifunctional hyaluronic acid-MnO2 nanoparticles in glioma. Adv Healthc Mater. 2019;8:1900047
154. Wei R, Gong X, Lin H, Zhang K, Li A, Liu K. et al. Versatile octapod-shaped hollow porous manganese (II) oxide nanoplatform for real-time visualization of cargo delivery. Nano Lett. 2019;19:5394-5402
155. Wu M, Hou P, Dong L, Cai L, Chen Z, Zhao M. et al. Manganese dioxide nanosheets: from preparation to biomedical applications. Int J nanomed. 2019;14:4781
156. Liu Y, Gong CS, Lin L, Zhou Z, Liu Y, Yang Z. et al. Core-shell metal-organic frameworks with fluorescence switch to trigger an enhanced photodynamic therapy. Theranostics. 2019;9:2791-2799
157. Ding B, Zheng P, Ma PA, Lin J. Manganese oxide nanomaterials: synthesis, properties, and theranostic applications. Adv Mater. 2020;32:1905823
158. Yi X, Chen L, Zhong X, Gao R, Qian Y, Wu F. et al. Core-shell Au@MnO2 nanoparticles for enhanced radiotherapy via improving the tumor oxygenation. Nano Res. 2016;9:3267-3278
159. Liang R, Liu L, He H, Chen Z, Han Z, Luo Z. et al. Oxygen-boosted immunogenic photodynamic therapy with gold nanocages@ manganese dioxide to inhibit tumor growth and metastases. Biomaterials. 2018;177:149-160
160. Liu J, Cui H, Yan S, Jing X, Wang D, Meng L. Gold nanostars decorated MnO2 nanosheets for magnetic resonance imaging and photothermal erasion of lung cancer cell. Mater Today Commun. 2018;16:97-104
161. Bi H, Dai Y, Yang P, Xu J, Yang D, Gai S. et al. Glutathione and H2O2 consumption promoted photodynamic and chemotherapy based on biodegradable MnO2-Pt@Au25 nanosheets. Chem Eng J. 2019;356:543-553
162. Fu D, Ding X, Wu J, Li C, Wang Q, Jiang J. Cationic polyelectrolyte mediated synthesis of MnO2-based core-shell structures as activatable MRI theranostic platform for tumor cell ablation. Part Part Syst Char. 2018;35:1800078
163. Ling Y, Zhang D, Cui X, Wei M, Zhang T, Wang J. et al. Direct monitoring of cell membrane vesiculation with 2D AuNP@MnO2 nanosheet supraparticles at the single-particle level. Angew Chem Int Edit. 2019;131:10652-10656
164. Yu J, Yang W, Xing S, Wang J, Han H, Zhang P. et al. Blended gold/MnO2@BSA nanoparticles for fluorometric and magnetic resonance determination of ascorbic acid. Microchim Acta. 2019;186:89
165. He W, Kim HK, Wamer WG, Melka D, Callahan JH, Yin JJ. Photogenerated charge carriers and reactive oxygen species in ZnO/Au hybrid nanostructures with enhanced photocatalytic and antibacterial activity. J Am Chem Soc. 2014;136:750-757
166. Deepagan VG, You DG, Um W, Ko H, Kwon S, Choi KY. et al. Long-circulating Au-TiO2 nanocomposite as a sonosensitizer for ROS-mediated eradication of cancer. Nano Lett. 2016;16:6257-6264
167. Lim DK, Barhoumi A, Wylie R, Reznor G, Langer R, Kohane DS. Enhanced photothermal effect of plasmonic nanoparticles coated with reduced graphene oxide. Nano Lett. 2013;13:4075-4079
168. Moon H, Kumar D, Kim H, Sim C, Chang JH, Kim JM. et al. Amplified photoacoustic performance and enhanced photothermal stability of reduced graphene oxide coated gold nanorods for sensitive photoacoustic imaging. ACS Nano. 2015;9:2711-2719
169. Yang X, Liu X, Liu Z, Pu F, Ren J, Qu X. Near-infrared light-triggered, targeted drug delivery to cancer cells by aptamer gated nanovehicles. Adv Mater. 2012;24:2890-2895
170. Ding X, Peng F, Zhou J, Gong W, Slaven G, Loh KP. et al. Defect engineered bioactive transition metals dichalcogenides quantum dots. Nat Commun. 2019;10:41
171. Peng F, Setyawati MI, Tee J K, Ding X, Wang J, Nga ME. et al. Nanoparticles promote in vivo breast cancer cell intravasation and extravasation by inducing endothelial leakiness. Nat Nanotechnol. 2019;14:279-286
172. Baptista PV. Cancer nanotechnology-prospects for cancer diagnostics and therapy. Curr Cancer Ther Rev. 2009;5:80-88
173. Hassan S, Prakash G, Ozturk AB, Saghazadeh S, Sohail MF, Seo J. et al. Evolution and clinical translation of drug delivery nanomaterials. Nano Today. 2017;15:91-106
174. Agrahari V, Agrahari V. Facilitating the translation of nanomedicines to a clinical product: challenges and opportunities. Drug Discov Today. 2018;23:974-991
175. Coty JB, Vauthier C. Characterization of nanomedicines: a reflection on a field under construction needed for clinical translation success. J Control Release. 2018;275:254-268
176. Ioannidis JP, Kim BY, Trounson A. How to design preclinical studies in nanomedicine and cell therapy to maximize the prospects of clinical translation. Nat Biomed Eng. 2018;2:797-809
177. Witzigmann D, Hak S, van der Meel R. Translating nanomedicines: thinking beyond materials? A young investigator's reply to 'the novelty bubble'. J Control Release. 2018;290:138-140
178. Goodman CM, McCusker CD, Yilmaz T, Rotello VM. Toxicity of gold nanoparticles functionalized with cationic and anionic side chains. Bioconj Chem. 2004;15:897-900
179. Carnovale C, Bryant G, Shukla R, Bansal V. Size, shape and surface chemistry of nano-gold dictate its cellular interactions, uptake and toxicity. Prog Mater Sci. 2016;83:152-190
180. Jia YP, Ma BY, Wei XW, Qian ZY. The in vitro and in vivo toxicity of gold nanoparticles. Chin Chem Lett. 2017;28:691-702
181. Ginzburg AL, Truong L, Tanguay RL, Hutchison JE. Synergistic toxicity produced by mixtures of biocompatible gold nanoparticles and widely used surfactants. ACS Nano. 2018;12:5312-5322
182. Conde J, Doria G, Baptista P. Noble metal nanoparticles applications in cancer. J Drug Deliv. 2012;2012:751075
183. Fang RH, Hu CMJ, Luk BT, Gao W, Copp JA, Tai Y. et al. Cancer cell membrane-coated nanoparticles for anticancer vaccination and drug delivery. Nano Lett. 2014;14:2181-2188
184. Wan Y, Wang L, Zhu C, Zheng Q, Wang G, Tong J. et al. Aptamer-conjugated extracellular nanovesicles for targeted drug delivery. Cancer Res. 2018;78:798-808
185. Han X, Shen S, Fan Q, Chen G, Archibong E, Dotti G. et al. Red blood cell-derived nanoerythrosome for antigen delivery with enhanced cancer immunotherapy. Sci Adv. 2019;5:eaaw6870
186. Jiang Y, Chekuri S, Fang RH, Zhang L. Engineering biological interactions on the nanoscale. Curr Opin Biotechnol. 2019;58:1-8
187. Zhou J, Kroll AV, Holay M, Fang RH, Zhang L. Biomimetic nanotechnology toward personalized vaccines. Adv Mater. 2019 p:1901255
188. Sindhwani S, Syed AM, Ngai J, Kingston BR, Maiorino L, Rothschild J. et al. The entry of nanoparticles into solid tumours. Nat Mater. 2020;19:566-575
Author contact
Corresponding author: Jiang Jiang, E-mail: jjiang2010ac.cn.
 Global reach, higher impact
Global reach, higher impact