Impact Factor
Theranostics 2020; 10(18):8130-8142. doi:10.7150/thno.47407 This issue Cite
Research Paper
Screening for in-vivo regional contractile defaults to predict the delayed Doxorubicin Cardiotoxicity in Juvenile Rat
1. PHYMEDEXP, INSERM, CNRS, Université de Montpellier, CHRU Montpellier, Montpellier, France.
2. FATH-IREC, Université Catholique de Louvain, Brussel, Belgium.
3. Pediatric and Congenital Cardiology Department, M3C Regional Reference CHD Centre, University Hospital, Montpellier, France.
4. Service d'onco-hématologie pédiatrique, CHRU Montpellier, Montpellier, France.
5. Unité de Pharmacie Clinique Oncologique, CHRU Montpellier, Montpellier, France.
*These authors contributed equally to this work.
Abstract
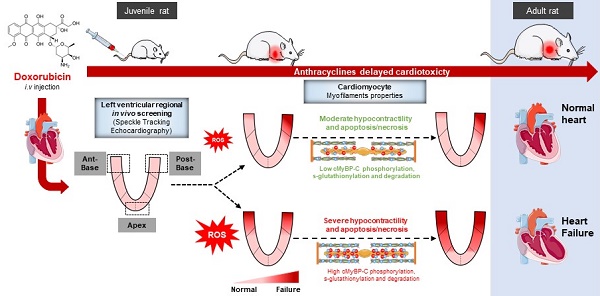
Anthracyclines are key chemotherapeutic agents used in various adult and pediatric cancers, however, their clinical use is limited due to possible congestive heart failure (HF) caused by acute and irreversible cardiotoxicity. Currently, there is no method to predict the future development of the HF in these patients. In order to identify early biomarkers to predict anthracycline cardiotoxicity in long-term survivors of childhood cancer, this longitudinal study aimed to analyze early and late in-vivo regional myocardial anthracycline-induced cardiotoxicity, related to in-vitro cardiac myocytes dysfunction, in a juvenile rat model.
Methods: Young male Wistar rats (4 weeks-old) were treated with different cumulative doses of doxorubicin (7.5, 10 or 12.5 mg/kg) or NaCl (0.9%) once a week for 6 weeks by intravenous injection. Cardiac function was evaluated in-vivo by conventional (left ventricular ejection fraction, LVEF) and regional two-dimensional (2D) speckle tracking echocardiography over the 4 months after the last injection. The animals were assigned to preserved (pEF) or reduced EF (rEF) groups at the end of the protocol and were compared to controls.
Results: We observed a preferential contractile dysfunction of the base of the heart, further altered in the posterior segment, even in pEF group. The first regional alterations appeared 1 month after chemotherapy. Functional investigation of cardiomyocytes isolated from the LV base 1 month after doxorubicin treatment showed that early in-vivo contractile alterations were associated with both decreased myofilament Ca2+ sensitivity and length-dependent activation. Changes in post-translational modifications (phosphorylation; S-glutathionylation) and protein degradation of the cardiac myosin binding protein-C may contribute to these alterations.
Conclusion: Our data suggest that screening of the contractile defaults of the base of the heart by regional 2D strain echocardiography is useful to detect subclinical myocardial dysfunction prior to the development of delayed anthracycline-induced cardiomyopathy in pediatric onco-cardiology.
Keywords: Anthracyclines, cardiotoxicity, regional speckle tracking echocardiography, heart failure, myofilament
 Global reach, higher impact
Global reach, higher impact