Impact Factor
Theranostics 2020; 10(18):8162-8178. doi:10.7150/thno.45088 This issue Cite
Research Paper
Morphological transformation enhances Tumor Retention by Regulating the Self-assembly of Doxorubicin-peptide Conjugates
1. College of pharmacy, Nanjing University of Chinese Medicine, Nanjing 210023, China.
2. Department of Pharmacy, the Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou 215002, China.
3. School of Traditional Chinese Pharmacy, China Pharmaceutical University, Nanjing 211198, China.
4. Key Laboratory of Xin'an Medicine, Ministry of Education, Anhui University of Chinese Medicine, Hefei, Anhui 230038, People's Republic of China.
*These authors contributed equally to this work.
Abstract
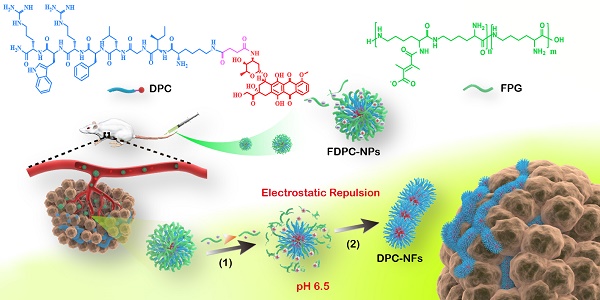
Rationale: Both spatial accuracy and temporal persistence are crucial in drug delivery, especially for anti-tumor intravenous nanomedicines, which have limited persistence due to their small particle sizes and easy removal from tumors. The present study takes advantage of morphological transformation strategy to regulate intravenous nanomedicines to display different sizes in different areas, achieving high efficient enrichment and long retention in lesions.
Methods: We designed and synthesized functional doxorubicin-peptide conjugate nanoparticles (FDPC-NPs) consisting of self-assembled doxorubicin-peptide conjugates (DPCs) and an acidic-responsive shielding layer named the functional polylysine graft (FPG), which can regulate the assembly morphology of the DPCs from spherical DPC nanoparticles (DPC-NPs) to DPC-nanofibers (DPC-NFs) by preventing the assembly force from π-π stacking and hydrogen bond between the DPC-NPs. The morphology transformation and particle changes of FDPC-NPs in different environments were determined with DLS, TEM and SEM. We used FRET to explore the enhanced retention effect of FDPC-NPs in tumor site in vivo. HPLC-MS/MS analytical method was established to analyze the biodistribution of FDPC-NPs in H22 hepatoma xenograft mouse model. Finally, the antitumor effect and safety of FDPC-NPs was evaluated.
Results: The FDPC-NPs were stable in blood circulation and responsively self-assembled into DPC-NFs when the FDPC-NPs underwent the acid-sensitive separation of the shielding layer in a mildly acidic microenvironment. The FDPC-NPs maintained a uniform spherical size of 80 nm and exhibited good morphological stability in neutral aqueous solution (pH 7.4) but aggregated into a long necklace-like chain structure or a crosslinked fiber structure over time in a weakly acidic solution (pH 6.5). These acidity-triggered transformable FDPC-NPs prolonged the accumulation in tumor tissue for more than 5 days after a single injection and improved the relative uptake rate of doxorubicin in tumors 31-fold. As a result, FDPC-NPs exhibited a preferable anti-tumor efficacy and a reduced side effect in vivo compared with free DOX solution and DOX liposomes.
Conclusions: Morphology-transformable FDPC-NPs represent a promising therapeutic approach for prolonging the residence time of drugs at the target site to reduce side effect and enhance therapeutic efficacy. Our studies provide a new and simple idea for the design of long-term delivery systems for intravenous chemotherapeutic drugs.
Keywords: intravenous nanomedicine, acid-responsive, morphology transformation, long retention, peptide-drug conjugates
 Global reach, higher impact
Global reach, higher impact