13.3
Impact Factor
Theranostics 2020; 10(20):9083-9099. doi:10.7150/thno.46467 This issue Cite
Research Paper
Lentinan-functionalized Selenium Nanoparticles target Tumor Cell Mitochondria via TLR4/TRAF3/MFN1 pathway
1. State Key Laboratory of Medicinal Chemical Biology and College of Pharmacy, Nankai University, Tianjin, China.
2. Tianjin Key Laboratory of Early Druggability Evaluation of Innovative Drugs and Tianjin Key Laboratory of Molecular Drug Research, Tianjin International Joint Academy of Biomedicine, Tianjin, China.
3. Department of Gastroenterology and Hepatology, General Hospital, Tianjin Medical University, Tianjin Institute of Digestive Disease, Tianjin, China.
4. Department of Anesthesiology, Tianjin Fourth Central Hospital, Tianjin, China.
*These authors have contributed equally to this work.
Abstract
Rationale: Malignant ascites caused by cancer cells results in poor prognosis and short average survival time. No effective treatment is currently available for malignant ascites. In this study, the effects of lentinan (LNT)-functionalized selenium nanoparticles (Selene) on malignant ascites were evaluated. Furthermore, the mechanism of Selene targeting mitochondria of tumor cells were also investigated.
Methods: Selene were synthesized and characterized by TEM, AFM and particle size analysis. The OVCAR-3 and EAC cells induced ascites models were used to evaluate the effects of Selene on malignant ascites. Proteomic analysis, immunofluorescence, TEM and ICP-MS were used to determine the location of Selene in tumor cells. Mitochondrial membrane potential, ROS, ATP content, and caspase-1/3 activity were detected to evaluate the effect of Selene on mitochondrial function and cell apoptosis. Immunofluorescence, Co-IP, pull-down, duolink, Western blot, and FPLC were used to investigate the pathway of Selene targeting mitochondria.
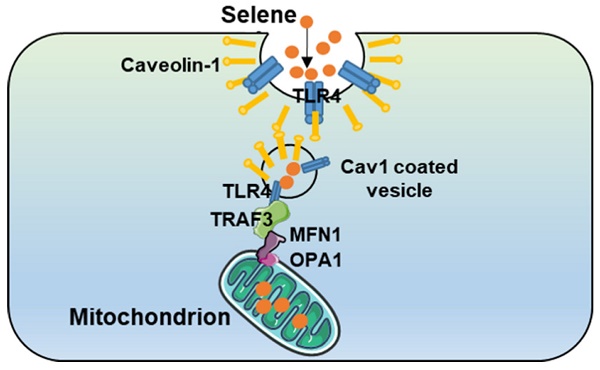
Results: Selene could effectively inhibit ascites induced by OVCAR-3 and EAC cells. Selene was mainly located in the mitochondria of tumor cells and induced apoptosis of tumor cells. The LNT in Selene was involved in caveolae-mediated endocytosis through the interaction between toll-like receptor-4 (TLR4) and caveolin 1 (CAV1). Furthermore, the Selene in the endocytic vesicles could enter the mitochondria via the mitochondrial membrane fusion pathway, which was mediated by TLR4/TNF receptor associated factor 3 (TRAF3)/mitofusin-1 (MFN1) protein complex.
Conclusion: Selene is a candidate anticancer drug for the treatment of malignant ascites. And TLR4/TRAF3/MFN1 may be a specific nano-drug delivery pathway that could target the mitochondria.
Keywords: malignant ascites, ovarian cancer, lentinan, selenium nanoparticles, mitochondria targeting pathway
 Global reach, higher impact
Global reach, higher impact