13.3
Impact Factor
Theranostics 2020; 10(20):9132-9152. doi:10.7150/thno.46076 This issue Cite
Research Paper
Tumor Microenvironment-triggered Nanosystems as dual-relief Tumor Hypoxia Immunomodulators for enhanced Phototherapy
1. Department of Pharmaceutics, School of Pharmacy, Qingdao University, Qingdao, 266021, China.
2. Department of Electrical and Computer Engineering, Tufts University, Medford, MA, 02155, USA.
3. Department of Pharmacology, School of Pharmacy, Qingdao University, Qingdao, 266021, China.
Abstract
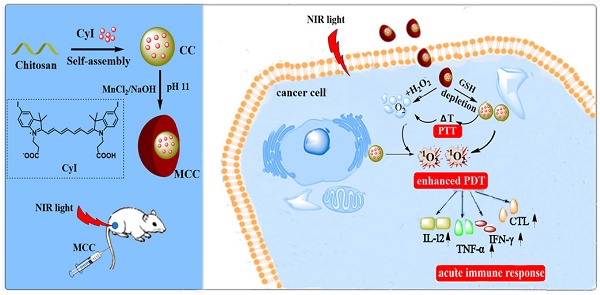
Photodynamic therapy (PDT) is a promising strategy in cancer treatment that utilizes photosensitizers (PSs) to produce reactive oxygen species (ROS) and eliminate cancer cells under specific wavelength light irradiation. However, special tumor environments, such as those with overexpression of glutathione (GSH), which will consume PDT-mediated ROS, as well as hypoxia in the tumor microenvironment (TME) could lead to ineffective treatment. Moreover, PDT is highly light-dependent and therefore can be hindered in deep tumor cells where light cannot easily penetrate. To solve these problems, we designed oxygen-dual-generating nanosystems MnO2@Chitosan-CyI (MCC) for enhanced phototherapy.
Methods: The TME-sensitive nanosystems MCC were easily prepared through the self-assembly of iodinated indocyanine green (ICG) derivative CyI and chitosan, after which the MnO2 nanoparticles were formed as a shell by electrostatic interaction and Mn-N coordinate bonding.
Results: When subjected to NIR irradiation, MCC offered enhanced ROS production and heat generation. Furthermore, once endocytosed, MnO2 could not only decrease the level of GSH but also serve as a highly efficient in situ oxygen generator. Meanwhile, heat generation-induced temperature increase accelerated in vivo blood flow, which effectively relieved the environmental tumor hypoxia. Furthermore, enhanced PDT triggered an acute immune response, leading to NIR-guided, synergistic PDT/photothermal/immunotherapy capable of eliminating tumors and reducing tumor metastasis.
Conclusion: The proposed novel nanosystems represent an important advance in altering TME for improved clinical PDT efficacy, as well as their potential as effective theranostic agents in cancer treatment.
Keywords: Tumor microenvironment, dual-relief hypoxia, enhanced PDT, acute immune response, synergistic anticancer therapy
 Global reach, higher impact
Global reach, higher impact