Impact Factor
Theranostics 2020; 10(20):9186-9199. doi:10.7150/thno.46825 This issue Cite
Research Paper
GYS1 induces glycogen accumulation and promotes tumor progression via the NF-κB pathway in Clear Cell Renal Carcinoma
1. Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou 510060, P. R. China.
2. Department of Pathology, Sun Yat-sen University Cancer Center, Guangzhou 510060, P. R. China.
#These authors have contributed equally to this work.
Abstract
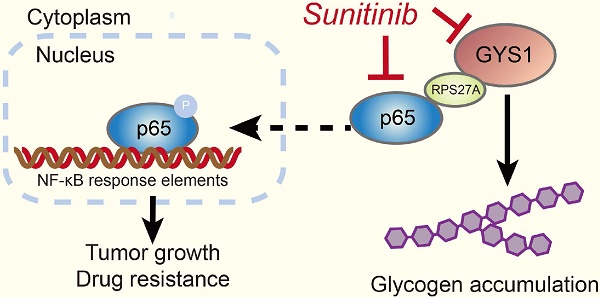
Metabolism reprogramming is a hallmark of many cancer types. We focused on clear cell renal carcinoma (ccRCC) which is characterized by its clear and glycogen-enriched cytoplasm with unknown reasons. The aim of this study was to identify the clinical significance, biological function, and molecular regulation of glycogen synthase 1 (GYS1) in ccRCC glycogen accumulation and tumor progression.
Methods: We determined the clinical relevance of GYS1 and glycogen in ccRCC by immunohistochemistry and periodic acid-schiff staining in fresh tissue and by tissue micro-array. Metabolic profiling with GYS1 depletion was performed by metabolomics analysis. In vitro and xenograft mouse models were used to evaluate the impact of GYS1 on cell proliferation. High-throughput RNA-Seq analyses and co-immunoprecipitation-linked mass spectrometry were used to investigate the downstream targets of GYS1. Flow cytometry and CCK8 assays were performed to determine the effect of GYS1 and sunitinib on cell viability.
Results: We observed that GYS1 was significantly overexpressed and glycogen was accumulated in ccRCC tissues. These effects were correlated with unfavorable patient survival. Silencing of GYS1 induced metabolomic perturbation manifested by a carbohydrate metabolism shift. Overexpression of GYS1 promoted tumor growth whereas its silencing suppressed it by activating the canonical NF-κB pathway. The indirect interaction between GYS1 and NF-κB was intermediated by RPS27A, which facilitated the phosphorylation and nuclear import of p65. Moreover, silencing of GYS1 increased the synthetic lethality of ccRCC cells to sunitinib treatment by concomitantly suppressing p65.
Conclusions: Our study findings reveal an oncogenic role for GYS1 in cell proliferation and glycogen metabolism in ccRCC. Re-sensitization of ccRCC cells to sunitinib suggests that GYS1 is a useful indicator of unfavorable prognosis as well as a therapeutic target for patients with ccRCC.
Keywords: clear cell renal carcinoma, GYS1, glycogen, metabolomics, NF-κB
 Global reach, higher impact
Global reach, higher impact