13.3
Impact Factor
Theranostics 2020; 10(20):9315-9331. doi:10.7150/thno.46875 This issue Cite
Research Paper
[18F]CB251 PET/MR imaging probe targeting translocator protein (TSPO) independent of its Polymorphism in a Neuroinflammation Model
1. Department of Nuclear Medicine, Seoul National University Hospital, Republic of Korea.
2. Biomedical Sciences, Cancer Research Institute, Seoul National University College of Medicine, Republic of Korea.
3. Laboratory of Molecular Imaging and Therapy, Cancer Research Institute, Seoul National University College of Medicine, Republic of Korea.
4. Department of Nuclear Medicine, Seoul National University Bundang Hospital, Republic of Korea.
5. Department of Transdisciplinary Studies, Graduate School of Convergence Science and Technology, Seoul National University, Republic of Korea.
6. Center for Nanomolecular Imaging and Innovative Drug Development, Advanced Institutes of Convergence Technology, Seoul National University, Republic of Korea.
7. Department of Neurosurgery, Seoul National University Hospital, Republic of Korea.
8. Brightonix Imaging Inc., National Cancer Institute, Republic of Korea.
9. Department of Nuclear Medicine, National Cancer Institute, Republic of Korea.
Abstract
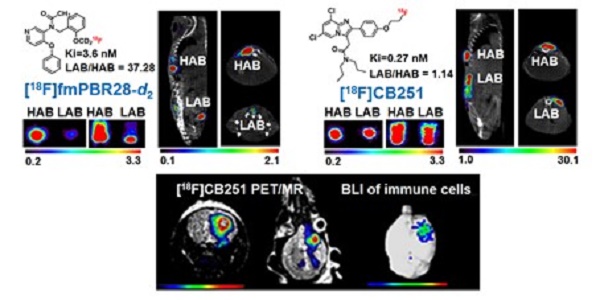
The 18 kDa translocator protein (TSPO) has been proposed as a biomarker for the detection of neuroinflammation. Although various PET probes targeting TSPO have been developed, a highly selective probe for detecting TSPO is still needed because single nucleotide polymorphisms in the human TSPO gene greatly affect the binding affinity of TSPO ligands. Here, we describe the visualization of neuroinflammation with a multimodality imaging system using our recently developed TSPO-targeting radionuclide PET probe [18F]CB251, which is less affected by TSPO polymorphisms.
Methods: To test the selectivity of [18F]CB251 for TSPO polymorphisms, 293FT cells expressing polymorphic TSPO were generated by introducing the coding sequences of wild-type (WT) and mutant (Alanine → Threonine at 147th Amino Acid; A147T) forms. Competitive inhibition assay was conducted with [3H]PK11195 and various TSPO ligands using membrane proteins isolated from 293FT cells expressing TSPO WT or mutant-A147T, representing high-affinity binder (HAB) or low-affinity binder (LAB), respectively. IC50 values of each ligand to [3H]PK11195 in HAB or LAB were measured and the ratio of IC50 values of each ligand to [3H]PK11195 in HAB to LAB was calculated, indicating the sensitivity of TSPO polymorphism. Cellular uptake of [18F]CB251 was measured with different TSPO polymorphisms, and phantom studies of [18F]CB251-PET using 293FT cells were performed. To test TSPO-specific cellular uptake of [18F]CB251, TSPO expression was regulated with pCMV-TSPO (or shTSPO)/eGFP vector. Intracranial lipopolysaccharide (LPS) treatment was used to induce regional inflammation in the mouse brain. Gadolinium (Gd)-DOTA MRI was used to monitor the disruption of the blood-brain barrier (BBB) and infiltration by immune cells. Infiltration of peripheral immune cells across the BBB, which exacerbates neuroinflammation to produce higher levels of neurotoxicity, was also monitored with bioluminescence imaging (BLI). Peripheral immune cells isolated from luciferase-expressing transgenic mice were transferred to syngeneic inflamed mice. Neuroinflammation was monitored with [18F]CB251-PET/MR and BLI. To evaluate the effects of anti-inflammatory agents on intracranial inflammation, an inflammatory cytokine inhibitor, 2-cyano-3, 12-dioxooleana-1, 9-dien-28-oic acid methyl ester (CDDO-Me) was administered in intracranial LPS challenged mice.
Results: The ratio of IC50 values of [18F]CB251 in HAB to LAB indicated similar binding affinity to WT and mutant TSPO and was less affected by TSPO polymorphisms. [18F]CB251 was specific for TSPO, and its cellular uptake reflected the amount of TSPO. Higher [18F]CB251 uptake was also observed in activated immune cells. Simultaneous [18F]CB251-PET/MRI showed that [18F]CB251 radioactivity was co-registered with the MR signals in the same region of the brain of LPS-injected mice. Luciferase-expressing peripheral immune cells were located at the site of LPS-injected right striatum. Quantitative evaluation of the anti-inflammatory effect of CDDO-Me on neuroinflammation was successfully monitored with TSPO-targeting [18F]CB251-PET/MR and BLI.
Conclusion: Our results indicate that [18F]CB251-PET has great potential for detecting neuroinflammation with higher TSPO selectivity regardless of polymorphisms. Our multimodal imaging system, [18F]CB251-PET/MRI, tested for evaluating the efficacy of anti-inflammatory agents in preclinical studies, might be an effective method to assess the severity and therapeutic response of neuroinflammation.
Keywords: neuroinflammation, TSPO, polymorphism, PET/MRI, multimodal imaging
 Global reach, higher impact
Global reach, higher impact