13.3
Impact Factor
Theranostics 2020; 10(20):9378-9394. doi:10.7150/thno.48247 This issue Cite
Research Paper
MED13L integrates Mediator-regulated epigenetic control into lung cancer radiosensitivity
1. Shandong Provincial Key Laboratory of Radiation Oncology, Cancer Research Center, Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences, Jinan, Shandong Province, 250117, China.
2. Department of Radiation Oncology, Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences, Jinan, Shandong Province, 250117, China.
3. Department of Radiation Oncology, Huaian No. 2 Hospital, Huaian, Jiangsu Province, 223002, China.
Abstract
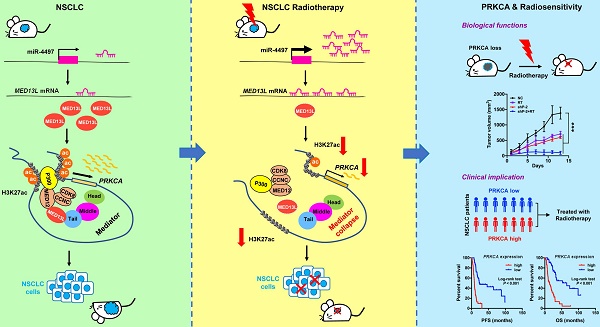
To date, efforts to improve non-small-cell lung cancer (NSCLC) outcomes with increased radiation dose have not been successful. Identification of novel druggable targets that are capable to modulate NSCLC radiosensitivity may provide a way forward. Mediator complex is implicated in gene expression control, but it remains unclear how Mediator dysfunction is involved in cancer radiotherapy.
Methods: The biologic functions of miR-4497, MED13L and PRKCA in NSCLC radiosensitivity were examined through biochemical assays including gene expression profilling, cell proliferation assay, colony formation assay, wound healing assay, transwell assay, dual luciferase reporter assay, xenograft models, immunoprecipitation, and chromatin immunoprecipitation sequencing. Clinical implications of miR-4497, MED13L and PRKCA in radiosensitivity were evaluated in NSCLC patients treated with concurrent chemoradiotherapy or radiotherapy alone.
Results: We found that radiation can trigger disassemble of Mediator complex via silencing of MED13L by miR-4497 in NSCLC. Although not interrupting structure integrity of the core Mediator or the CDK8 kinase module, suppression of MED13L attenuated their physical interactions and reduced recruitment of acetyltransferase P300 to chromatin via Mediator. Silencing of MED13L therefore diminishes global H3K27ac signals written by P300, activities of enhancer and/or promoters and expression of multiple oncogenes, especially PRKCA. Inhibition of PRKCA expression potentiates the killing effect of radiotherapy in vitro and in vivo. Remarkably, high PRKCA expression in NSCLC tissues is correlated with poor prognosis of patients received radiotherapy.
Conclusions: Our study linking PRKCA to radiosensitivity through a novel mechanism may enable the rational targeting of PRKCA to unlock therapeutic potentials of NSCLC.
Keywords: lung cancer radiotherapy, MED13L, miR-4497, Mediator, H3K27ac, PRKCA
 Global reach, higher impact
Global reach, higher impact