Impact Factor
Theranostics 2020; 10(21):9512-9527. doi:10.7150/thno.44394 This issue Cite
Research Paper
Pharmacological depletion of microglia and perivascular macrophages prevents Vascular Cognitive Impairment in Ang II-induced hypertension
1. Department of Neurology, Maastricht University Medical Center, 6202AZ, Maastricht, The Netherlands.
2. Department of Pathology, Maastricht University, Maastricht, 6200MD, The Netherlands.
3. Department of Psychiatry and Neuropsychology, Maastricht University, 6200MD, Maastricht, The Netherlands.
4. Department of Pharmacology and Toxicology, Maastricht University, 6200MD, Maastricht, The Netherlands.
5. CARIM - School for Cardiovascular Diseases, Maastricht University, 6200MD, The Netherlands.
6. MH&Ns - School for Mental Health and Neuroscience, Maastricht University, 6200MD, The Netherlands.
7. IMCAR - Institute for Molecular Cardiology Research, RWTH Aachen, 52074, Germany.
Abstract
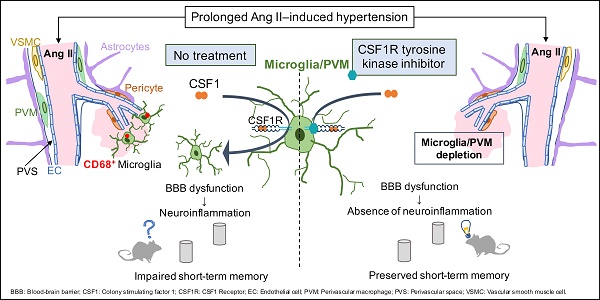
Rationale: Hypertension is a major risk factor for cerebral small vessel disease, the most prevalent cause of vascular cognitive impairment. As we have shown, hypertension induced by a prolonged Angiotensin II infusion is associated with increased permeability of the blood-brain barrier (BBB), chronic activation of microglia and myelin loss. In this study we therefore aim to determine the contribution of microglia to hypertension-induced cognitive impairment in an experimental hypertension model by a pharmacological depletion approach.
Methods: For this study, adult Cx3Cr1gfp/wtxThy1yfp/0 reporter mice were infused for 12 weeks with Angiotensin II or saline and subgroups were treated with PLX5622, a highly selective CSF1R tyrosine kinase inhibitor. Systolic blood pressure (SBP) was measured via tail-cuff. Short- and long-term spatial memory was assessed during an Object Location task and a Morris Water Maze task (MWM). Microglia depletion efficacy was assessed by flow cytometry and immunohistochemistry. BBB leakages, microglia phenotype and myelin integrity were assessed by immunohistochemistry.
Results: SBP, heart weight and carotid pulsatility were increased by Ang II and were not affected by PLX5622. Short-term memory was significantly impaired in Ang II hypertensive mice, and partly prevented in Ang II mice treated with PLX5622. Histological and flow cytometry analysis revealed almost complete ablation of microglia and a 60% depletion of brain resident perivascular macrophages upon CSF1R inhibition. Number and size of BBB leakages were increased in Ang II hypertensive mice, but not altered by PLX5622 treatment. Microglia acquired a pro-inflammatory phenotype at the site of BBB leakages in both Saline and Ang II mice and were successfully depleted by PLX5622. There was however no significant change in myelin integrity at the site of leakages.
Conclusion: Our results show that depletion of microglia and PVMs, by CSF1R inhibition prevents short-term memory impairment in Ang II induced hypertensive mice. We suggest this beneficial effect is mediated by the major decrease of pro-inflammatory microglia within BBB leakages. This novel finding supports the critical role of brain immune cells in the pathogenesis of hypertension-related cognitive impairment. An adequate modulation of microglia /PVM density and phenotype may constitute a relevant approach to prevent and/or limit the progression of vascular cognitive impairment.
 Global reach, higher impact
Global reach, higher impact