Impact Factor
Theranostics 2020; 10(21):9686-9701. doi:10.7150/thno.44146 This issue Cite
Research Paper
Retinoic acid promotes metabolic maturation of human Embryonic Stem Cell-derived Cardiomyocytes
1. Department of Cardiovascular Surgery of the First Affiliated Hospital & Institute for Cardiovascular Science, Collaborative Innovation Center of Hematology, State Key Laboratory of Radiation Medicine and Protection, Medical College, Soochow University, Suzhou 215000, China.
2. State Key Laboratory of Stem Cell and Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences, Beijing 100101, China.
3. University of Chinese Academy of Sciences, Beijing 100049, China.
4. State Key Laboratory of Space Medicine Fundamentals and Application, China Astronaut Research and Training Center, Beijing 100094, China.
5. Fuwai Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100037, China.
6. Institute of Microcirculation & Department of Pathophysiology of Basic Medical College, Hebei North University, Zhangjiakou 075000, China.
*These authors contributed equally to this work.
Abstract
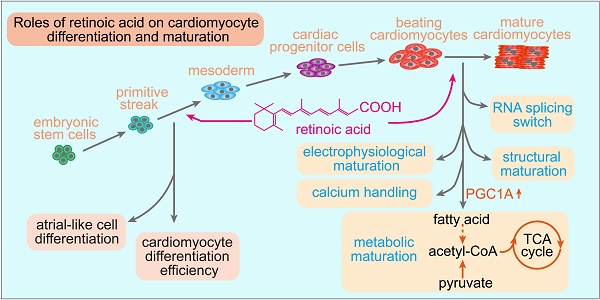
Cardiomyocytes differentiated from human embryonic stem cells (hESCs) represent a promising cell source for heart repair, disease modeling and drug testing. However, improving the differentiation efficiency and maturation of hESC-derived cardiomyocytes (hESC-CMs) is still a major concern. Retinoic acid (RA) signaling plays multiple roles in heart development. However, the effects of RA on cardiomyocyte differentiation efficiency and maturation are still unknown.
Methods: RA was added at different time intervals to identify the best treatment windows for cardiomyocyte differentiation and maturation. The efficiency of cardiomyocyte differentiation was detected by quantitative real-time PCR and flow cytometry. Cardiomyocytes maturation was detected by immunofluorescence staining, metabolic assays and patch clamp to verify structural, metabolic and electrophysiological maturation, respectively. RNA sequencing was used for splicing analysis.
Results: We found that RA treatment at the lateral mesoderm stage (days 2-4) significantly improved cardiomyocyte differentiation, as evidenced by the upregulation of TNNT2, NKX2.5 and MYH6 on day 10 of differentiation. In addition, flow cytometry showed that the proportion of differentiated cardiomyocytes in the RA-treated group was significantly higher than that in control group. RA treatment on days 15-20 increased cardiomyocyte area, sarcomere length, multinucleation and mitochondrial copy number. RNA sequencing revealed RA promoted RNA isoform switch to the maturation-related form. Meanwhile, RA promoted electrophysiological maturation and calcium handling of hESC-CMs. Importantly, RA-treated cardiomyocytes showed decreased glycolysis and enhanced mitochondrial oxidative phosphorylation, with the increased utilization of fatty acid and exogenous pyruvate but not glutamine.
Conclusion: Our data indicated that RA treatment at an early time window (days 2-4) promotes the efficiency of cardiomyocyte differentiation and that RA treatment post beating (days 15-20) promotes cardiomyocyte maturation. The biphasic effects of RA provide new insights for improving cardiomyocyte differentiation and quality.
Keywords: Embryonic stem cells, Cardiomyocyte differentiation, Retinoic acid, Cardiomyocyte maturation, Oxidative phosphorylation
 Global reach, higher impact
Global reach, higher impact