13.3
Impact Factor
Theranostics 2020; 10(21):9830-9842. doi:10.7150/thno.45161 This issue Cite
Research Paper
O-GlcNAcylation of SIX1 enhances its stability and promotes Hepatocellular Carcinoma Proliferation
1. State key Laboratory of Cancer Biology and National Clinical Research Center for Digestive Diseases, Xijing Hospital of Digestive Diseases, Fourth Military Medical University, Xi'an, 710032, China.
2. Lab of Tissue Engineering, Faculty of Life Science, Northwest University, Xi'an, 710032, China.
3. Department of Gastroenterology, Sixth Medical Center of PLA General Hospital, Beijing, 100048, China.
*These authors contributed equally to the paper.
Abstract
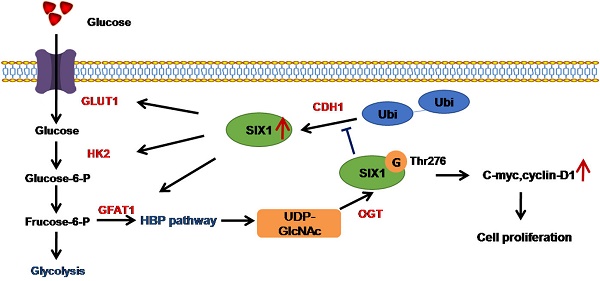
It is universally accepted that aberrant metabolism facilitates tumor growth. However, how cancer cells coordinate glucose metabolism and tumor proliferation is largely unknown. Sine oculis homeobox homolog 1 (SIX1) is a transcription factor that belongs to the SIX family and is believed to play an important role in the regulation of the Warburg effect in tumors. However, whether the role of SIX1 and the molecular mechanisms that regulate its activity are similar in hepatocellular carcinoma (HCC) still needs further investigation.
Methods: Western blotting was performed to determine the levels of SIX1 and O-linked β-N-acetylglucosaminylation (O-GlcNAcylation) in HCC tissues. Cell Counting Kit 8 (CCK8), colony formation and mouse tumor model assays were used to establish the role of SIX1 and O-GlcNAcylation in HCC processes. Mass spectrometry, immunoprecipitation and site-directed mutagenesis were performed to confirm the O-GlcNAcylation of SIX1.
Results: Here, we demonstrated that SIX1, the key transcription factor regulating the Warburg effect in cancer, promotes HCC growth in vitro and in vivo. Furthermore, we revealed that SIX1 could also enhance the levels of a posttranslational modification called O-GlcNAcylation. Importantly, we found that SIX1 was also highly modified by O-GlcNAcylation and that O-GlcNAcylation inhibited the ubiquitination degradation of SIX1. In addition, site-directed mutagenesis at position 276 (T276A) decreased the O-GlcNAcylation level and reversed the protumor effect of SIX1.
Conclusions: We conclude that O-GlcNAcylation of SIX1 enhances its stability and promotes HCC proliferation. Our findings illustrate a novel feedback loop of SIX1 and O-GlcNAcylation and show that O-GlcNAcylation of SIX1 is an important way to coordinate glucose metabolism and tumor progression.
Keywords: SIX1, O-GlcNAcylation, hepatocellular carcinoma, Warburg effect, ubiquitination
 Global reach, higher impact
Global reach, higher impact