13.3
Impact Factor
Theranostics 2020; 10(21):9865-9887. doi:10.7150/thno.46771 This issue Cite
Research Paper
Targeted Manganese doped silica nano GSH-cleaner for treatment of Liver Cancer by destroying the intracellular redox homeostasis
1. College of Pharmaceutical Sciences, Zhejiang Chinese Medical University, Hangzhou, 311400, China.
2. Libraries of Zhejiang Chinese Medical University, Zhejiang Chinese Medical University, Hangzhou, 310053, China.
#These authors equally contributed to this work.
Abstract
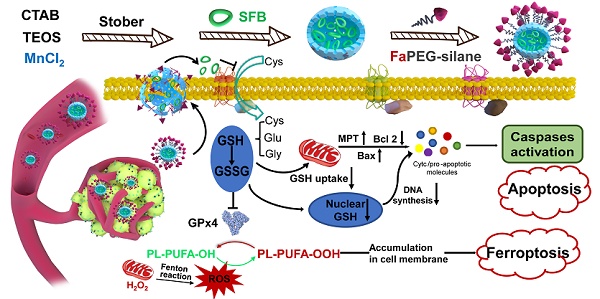
Background: Glutathione (GSH), the primary antioxidant in cells, could fight against oxidative stress. Tumor cells display a higher GSH level than normal cells for coping with the hyperoxidative state, which meets the requirements of enhanced metabolism and vicious proliferation. Therefore, the consumption of GSH will lead to cell redox imbalance and impede life activities. Herein, targeted sorafenib (SFB) loaded manganese doped silica nanoparticle (FaPEG-MnMSN@SFB) was constructed, which could destroy the intracellular redox homeostasis by consuming GSH.
Methods: In this study, MnMSN was prepared by an optimized one-pot Stober's method for loading SFB, and FaPEG chain was modified on the surface of MnMSN to achieve long circulation and targeted delivery. The anticancer efficacy and mechanism of the designed FaPEG-MnMSN@SFB were assessed both in vitro and in vivo.
Results: FaPEG-MnMSN@SFB exhibited efficient antitumor activity by dual depleting intracellular GSH (the degradation of MnMSN would consume intracellular GSH and the SFB would inhibit the effect of Xc- transport system to inhibit GSH synthesis). Moreover, disruption of redox balance would lead to apoptosis and reactive oxygen species (ROS)-dependent ferroptosis of tumor cells.
Conclusion: Such a GSH-starvation therapeutic strategy would cause multi-path programmed cell death and could be a promising strategy for cancer therapy.
Keywords: glutathione, nanocleaner, sorafenib, ferroptosis, apoptosis
 Global reach, higher impact
Global reach, higher impact