Impact Factor
Theranostics 2021; 11(1):1-13. doi:10.7150/thno.47435 This issue Cite
Research Paper
IL-37 exerts therapeutic effects in experimental autoimmune encephalomyelitis through the receptor complex IL-1R5/IL-1R8
1. Institut de Neurociencies and Departament de Biologia Cel·lular, Fisiologia i Immunologia, Universitat Autonoma de Barcelona, Bellaterra, Catalonia, Spain.
2. Centro de Investigación Biomédica en Red de Enfermedades Neurodegenerativas (CIBERNED).
3. Department of Neuroscience, Faculty of Medicine, Université de Montréal, and Neuroimmunology Unit, Centre de Recherche du CHUM (CRCHUM), Montréal, Québec, Canada.
4. Humanitas Clinical and Research Center, 20089 Rozzano, Italy.
5. Department of Biochemistry and Molecular Genetics, University of Colorado Denver, Aurora, CO 80238, USA.
6. Department of Medicine, University of Colorado Denver, Aurora, CO 80045, USA.
7. Department of Medicine, Radboud University Medical Center, 6500 Nijmegen, The Netherlands.
Abstract
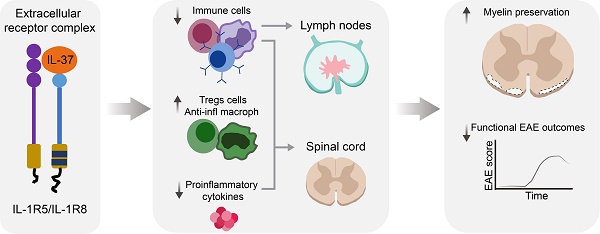
Background: Interleukin 37 (IL-37), a member of IL-1 family, broadly suppresses inflammation in many pathological conditions by acting as a dual-function cytokine in that IL-37 signals via the extracellular receptor complex IL1-R5/IL-1R8, but it can also translocate to the nucleus. However, whether IL-37 exerts beneficial actions in neuroinflammatory diseases, such as multiple sclerosis, remains to be elucidated. Thus, the goals of the present study were to evaluate the therapeutic effects of IL-37 in a mouse model of multiple sclerosis, and if so, whether this is mediated via the extracellular receptor complex IL-1R5/IL-1R8.
Methods: We used a murine model of MS, the experimental autoimmune encephalomyelitis (EAE). We induced EAE in three different single and double transgenic mice (hIL-37tg, IL-1R8 KO, hIL-37tg-IL-1R8 KO) and wild type littermates. We also induced EAE in C57Bl/6 mice and treated them with various forms of recombinant human IL-37 protein. Functional and histological techniques were used to assess locomotor deficits and demyelination. Luminex and flow cytometry analysis were done to assess the protein levels of pro-inflammatory cytokines and different immune cell populations, respectively. qPCRs were done to assess the expression of IL-37, IL-1R5 and IL-1R8 in the spinal cord of EAE, and in blood peripheral mononuclear cells and brain tissue samples of MS patients.
Results: We demonstrate that IL-37 reduces inflammation and protects against neurological deficits and myelin loss in EAE mice by acting via IL1-R5/IL1-R8. We also reveal that administration of recombinant human IL-37 exerts therapeutic actions in EAE mice. We finally show that IL-37 transcripts are not up-regulated in peripheral blood mononuclear cells and in brain lesions of MS patients, despite the IL-1R5/IL-1R8 receptor complex is expressed.
Conclusions: This study presents novel data indicating that IL-37 exerts therapeutic effects in EAE by acting through the extracellular receptor complex IL-1R5/IL-1R8, and that this protective physiological mechanism is defective in MS individuals. IL-37 may therefore represent a novel therapeutic avenue for the treatment of MS with great promising potential.
Keywords: multiple sclerosis, experimental autoimmune encephalomyelitis, IL-37, IL-1R8, cytokines
 Global reach, higher impact
Global reach, higher impact