13.3
Impact Factor
Theranostics 2021; 11(1):107-116. doi:10.7150/thno.50486 This issue Cite
Research Paper
Facile engineering of silk fibroin capped AuPt bimetallic nanozyme responsive to tumor microenvironmental factors for enhanced nanocatalytic therapy
1. State Key Laboratory of Silkworm Genome Biology, School of Materials and Energy, Southwest University, Chongqing 400715, China.
2. Cancer Center, Medical Research Institute, Southwest University, Chongqing 400716, China.
3. Chongqing Key Laboratory of Ultrasound Molecular Imaging, Institute of Ultrasound Imaging, Second Affiliated Hospital, Chongqing Medical University, Chongqing, 400010, China.
Abstract
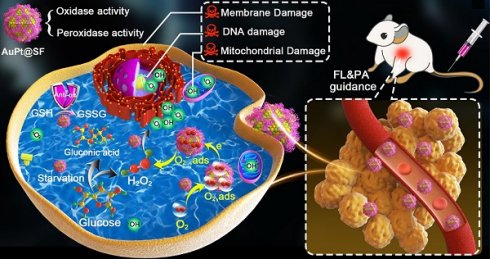
Background: Reactive oxygen species (ROS), as a category of highly reactive molecules, are attractive for eliminating tumor cells in situ. However, the intrinsic tumor microenvironment (TME) always compromises treatment efficacy. In another aspect, silk fibroin (SF), as a category of natural biomacromolecules, is highly promising for synthesis of metallic nanocrystals via biomineralization.
Methods: As a proof-of-concept study, AuPt bimetallic nanozyme derived from bioinspired crystallization of chloroauric acid and chloroplatinic acid was facilely developed in the presence of silk fibroin (SF). Antitumor effects caused by the as-synthesized AuPt@SF (APS) nanozyme were demonstrated in 4T1 tumor cells in vitro and xenograft tumor models in vivo.
Results: APS nanozyme can decompose glucose to constantly supply H2O2 and deplete intracellular glutathione (GSH). APS nanozyme can simultaneously convert adsorbed O2 and endogenic H2O2 into superoxide radicals (•O2-) and hydroxyl radical (•OH), respectively, upon highly efficient catalytic reaction. Subsequently, these cytotoxic ROS cause irreversible damage to the cell membrane, nucleic acid and mitochondria of tumors. Upon fluorescence/photoacoustic (FL/PA)-imaging guidance, remarkable tumor damage based on the current nanoplatform was confirmed in vivo.
Conclusion: The objective of our investigation is to supply more useful insights on the development of SF-based nanocatalysts, which are specifically responsive to TME for extremely efficient tumor theranostics.
Keywords: Silk fibroin, Nanocatalyst, Tumor theranostics, Biomineralization, Tumor microenvironment
 Global reach, higher impact
Global reach, higher impact