Impact Factor
Theranostics 2021; 11(1):316-329. doi:10.7150/thno.49713 This issue Cite
Review
Immunopathogenesis and treatment of cytokine storm in COVID-19
1. Department of Nephrology, Yonsei University Wonju College of Medicine, Wonju, Republic of Korea.
2. Department of Pediatrics, Yonsei University College of Medicine, Seoul, Republic of Korea.
3. Department of Internal Medicine I, Gastroenterology, Hepatology, Endocrinology & Metabolism, Medical University Innsbruck, Innsbruck, Austria.
4. Department of Nephrology, Rigshospitalet, University of Copenhagen, Copenhagen, Denmark.
5. Department of Internal Medicine IV, Nephrology and Hypertension, Medical University Innsbruck, Innsbruck, Austria.
Received 2020-6-19; Accepted 2020-9-20; Published 2021-1-1
Abstract
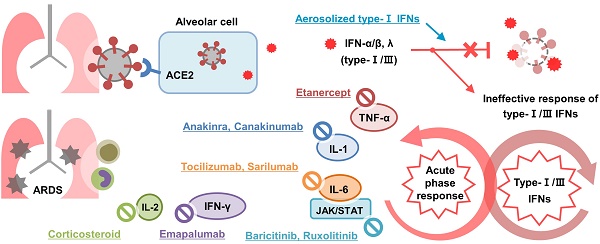
Severe coronavirus disease 2019 (COVID-19) is characterized by systemic hyper-inflammation, acute respiratory distress syndrome, and multiple organ failure. Cytokine storm refers to a set of clinical conditions caused by excessive immune reactions and has been recognized as a leading cause of severe COVID-19. While comparisons have been made between COVID-19 cytokine storm and other kinds of cytokine storm such as hemophagocytic lymphohistiocytosis and cytokine release syndrome, the pathogenesis of cytokine storm has not been clearly elucidated yet. Recent studies have shown that impaired response of type-1 IFNs in early stage of COVID-19 infection played a major role in the development of cytokine storm, and various cytokines such as IL-6 and IL-1 were involved in severe COVID-19. Furthermore, many clinical evidences have indicated the importance of anti-inflammatory therapy in severe COVID-19. Several approaches are currently being used to treat the observed cytokine storm associated with COVID-19, and expectations are especially high for new cytokine-targeted therapies, such as tocilizumab, anakinra, and baricitinib. Although a number of studies have been conducted on anti-inflammatory treatments for severe COVID-19, no specific recommendations have been made on which drugs should be used for which patients and when. In this review, we provide an overview of cytokine storm in COVID-19 and treatments currently being used to address it. In addition, we discuss the potential therapeutic role of extracorporeal cytokine removal to treat the cytokine storm associated with COVID-19.
Keywords: Coronavirus, COVID-19, cytokine storm, cytokine blockades, plasma exchange
Introduction
Coronavirus disease 2019 (COVID-19) has affected the world in a devastating way since December 2019. The causative virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has gained novel properties through variations in its original form. It now has great infectivity and is often fatal, causing acute respiratory distress syndrome (ARDS) and multiple organ failure [1]. In serious cases, clinical deterioration is often rapid, and in a large proportion the severe disease course is caused by systemic hyper-inflammation, the so called “cytokine storm” [2].
Cytokine storm is a condition of uncontrolled systemic hyper-inflammation caused by cytokine excess, leading to multi-organ failure and even death [3]. The concept of cytokine storm was initially recognized in acute graft-versus-host disease in the process of hematopoietic stem cell transplantation [4]. Researchers have since revealed that cytokine storm occurs in various diseases, such as malignancy, rheumatologic disease, and sepsis syndrome and is a concern in COVID-19 [5]. Even though a variety of treatments to mitigate cytokine storm have been introduced, no concrete treatment recommendations have been issued so far for COVID-19: in recent studies novel cytokine blockades targeting specific cytokines, such as interleukin (IL)-1 and IL-6 or Janus kinase (JAK) pathway have shown promising potential for the treatment of COVID-19. In addition, traditional anti-inflammatory drugs, including corticosteroids and colchicine, are also being explored for COVID-19 through active and vibrant researches [6].
In this study, we provide an overview of the cytokine storm caused by different etiologies and specific treatments thereof, focusing particularly on novel cytokine blockades. In addition, we discuss the pathophysiology and individual treatments for the cytokine storm in severe COVID-19. Last, we propose the potential of therapeutic plasma exchange (TPE) to treat cytokine storm in severe COVID-19. We believe that TPE is able to effectively remove inflammatory cytokines from the blood and can improve cytokine storm [7].
Immunopathogenesis of cytokine storm
Immune features of cytokine storm
Unpredicted adverse events in the phase 1 trial of the anti-CD28 monoclonal antibody TGN1412 provided clues about cytokine storm [8]. In just a few hours after administration of the study medication, participants experienced serious complications from cytokine storm. The patients all commonly experienced sudden fever, respiratory and kidney failure, hypotensive shock, and diffuse coagulation disorders. Laboratory tests revealed anemia, neutrophilia, thrombocytopenia, and marked lymphopenia. These clinical features of multi-organ failure are believed to be caused by activation of the following four cascades: complement, kinin, clotting, and fibrinolysis systems [9]. In addition, acute increases in various cytokines were also noted in the early phase: tumor necrosis factor-alpha (TNF-α) was observed within an hour, followed by IL-1 [8].
Since then, through experience with various clinical diseases, including hemophagocytic lymphohistiocytosis (HLH), the concept that an excessive immune response can seriously damage the body and causes rapid clinical deterioration began to be established [10]. Primary or familial HLH by genetic mutations and secondary HLH by specific viral infections, malignancies, autoimmune disorders, and iatrogenic causes are typical diseases accompanied by cytokine storm [11]. In particular, cytokine storm caused by autoimmune disorders is historically called macrophage activation syndrome (MAS) [12], and cytokine release syndrome (CRS) refers to the cytokine storm that occurs after chimeric antigen receptor (CAR) T cell therapy [13]. Sepsis is also an important cause for the development of cytokine storm, but it is not easy to discriminate cytokine storm from the clinical course of severe sepsis itself [14].
A variety of cytokines, including the IL-1 family, IL-6, IL-8, IL-10, TNF-α, and interferon (IFN)-γ, are involved in the development of a cytokine storm, although the key pathogenic cytokines appear to differ depending on the disease. IFN-γ is a key cytokine in primary HLH [15]. IL-1ß plays a major role in systemic juvenile idiopathic arthritis (SJIA) [16], while in MAS associated with SJIA, IL-18 may be a key cytokine [17]. Another representative cytokine, IL-6, is a key inflammatory molecule in CRS [18]. Importantly, even if the clinical symptoms caused by a cytokine storm exhibit a common pattern, treatment must be individualized because the degree to which each cytokine contributes to the development of the disease can differ [3]. On the other hand, cytokine storm in sepsis involves multiple factors, and thus, multi-directional suppression of inflammatory reactions is necessary.
Interestingly, leukocytopenia, particularly lymphopenia, has been found to be a typical finding in cytokine storm and to be associated with the severity thereof. One study has described a possible mechanism for lymphopenia in cytokine storm. After the TGN1412 trial, Muller et al. administered another anti-CD28 super-agonistic antibody, JJ316, to experimental rats and observed responses [19]. Similar to those that occurred in the TGN1412 trial, including lymphopenia. Further investigations revealed that T cells were rapidly redistributed to secondary lymphoid organs, such as the spleen and lymph nodes, and were trapped with decreased mobility. Although we are not sure why these findings occur as a part of systemic hyper-inflammatory reactions, considering that lymphopenia was seen without infection in the trial, it is not likely that lymphopenia is induced directly by a pathogenic organism, such as a virus. Instead, we believe that lymphopenia might be the result of effective immune reactions against severe systemic infections for more efficient trafficking of immune cells to secondary lymphoid organs. One other notable thing from the TGN1412 trial is that the violent inflammatory responses were followed by reactive immunosuppression. The responses, called “second wave” or “immune paralysis,” were found to downregulate the previous inflammatory reactions, which is likely why TGN1412 was suspected as showing therapeutic effects on autoimmune disease in preclinical studies in rats [20].
Research has yet to reveal whether cytokine storm results from abnormal innate or adaptive immunity. As observed in the TGN1412 trial, rapid onset of cytokine storm after drug administration and a series of inflammatory responses following an acute increase of TNF-α suggests a problem with innate immunity [8]. However, considering the elevation of soluble interleukin-2 receptor in secondary HLH [21] and the effects of standard treatment for HLH targeting dysregulated T cells [22], the problem of adaptive immunity is also thought to be the main mechanism of cytokine storm. Among various cytokines related to cytokine storm, IL-1 and IL-6 have been described as key cytokines in some patients. These cytokines are representative pro-inflammatory cytokines, along with TNF-α, that play an important role in acute phase responses of inflammation. Pattern recognition receptors (PRRs), such as toll-like receptor (TLR) and retinoic acid-inducible gene-1-like receptor (RLR), are proteins that present in the cytoplasm or cell membrane to recognize infection or tissue damage by combining with pathogen-associated molecular pattern (PAMP) derived from microorganisms or damage-associated molecular pattern (DAMP) resulting from tissue damage. IL-1, IL-6, and TNF-α are produced by dendritic cell and mononuclear macrophage that are activated by PRRs, and initiate a series of acute inflammatory reactions through activation of innate immunity [23]. Meanwhile, however, IL-6, along with IFN-γ, is also important for T-cell mediated adaptive immunity [24]. Therefore, dysregulated activation of T cells can be considered as a major pathological mechanism in a cytokine storm wherein IL-6 and IFN-γ are key cytokines. Taken together, although the clinical characteristics of cytokine storm are similar, underlying pathological mechanisms affecting the clinical course, prognosis, and treatment response in individual forms of cytokine storm can differ. Thus, it is important to find the key cytokine in various cytokine storms and to take a proper therapeutic approach by determining whether the instance is related with innate or adaptive immunity.
Cytokine storm in severe COVID-19
Studies have indicated that the rapid clinical deterioration and high mortality risk in severe COVID-19 could be related to cytokine storm [6]. One study showed that blood levels for various cytokines, such as IL-1ß, IFN-γ, IFN-γ-induced protein 10 (IP10), and monocyte chemoattractant protein 1 (MCP1), were elevated in COVID-19 [25]. In addition, patients admitted to the intensive care unit (ICU) had higher cytokine levels of IL-2, IL-7, IL-10, granulocyte colony-stimulating factor (G-CSF), IP10, MCP1, macrophage inflammatory protein 1- α, and TNF-α than those not requiring ICU treatment [25]. Another study showed that IL-6 was more elevated in non-survivors than survivors from COVID-19, suggesting that COVID-19 mortality might be due to virus-activated cytokine storm [26].
A recent article presented an interesting concept of a cytokine storm in COVID-19. The study argued that the cytokine storm in COVID-19 is the result from a failure of the immune system to remove the virus. They divided the cytokine storm into two stages [27]: The first stage is a temporary immune-deficient condition that is similar to primary HLH. The subsequent secondary stage is an overactive immune state to compensate for the target clearance failure, which appears as a clinical manifestation of a cytokine storm. Another review article also described the cytokine storm associated with COVID-19 in a similar context [28]. Cell and animal experiments investigating the effects of human coronavirus on cytokines have demonstrated delayed secretion of type Ⅰ and Ⅲ IFNs, including IFN α/ ß, in the early phase of infection and excessive secretion of pro-inflammatory cytokines from mononuclear macrophages in the later stage [29]. Another study in which immune analysis was performed in COVID-19 patients highlighted profound impaired type 1 IFN responses characterized by a low level of IFN activity and downregulation of IFN stimulated genes. In addition, the study reported hyper-inflammatory responses represented by IL-6 and TNF-α [30]. Taken together, these studies emphasize that a failure in initial type-Ⅰ and Ⅲ IFN responses to SARS-CoV-2 leads to an excessive late immune response and severe form of COVID-19 (Figure 1). In this respect we could figure out why severe COVID-19 is commonly accompanied by ARDS. The failure of immune response in initial period of SARS-CoV-2 infection induces generalized hyper-inflammation in lung that leads to acute lung injury and ARDS.
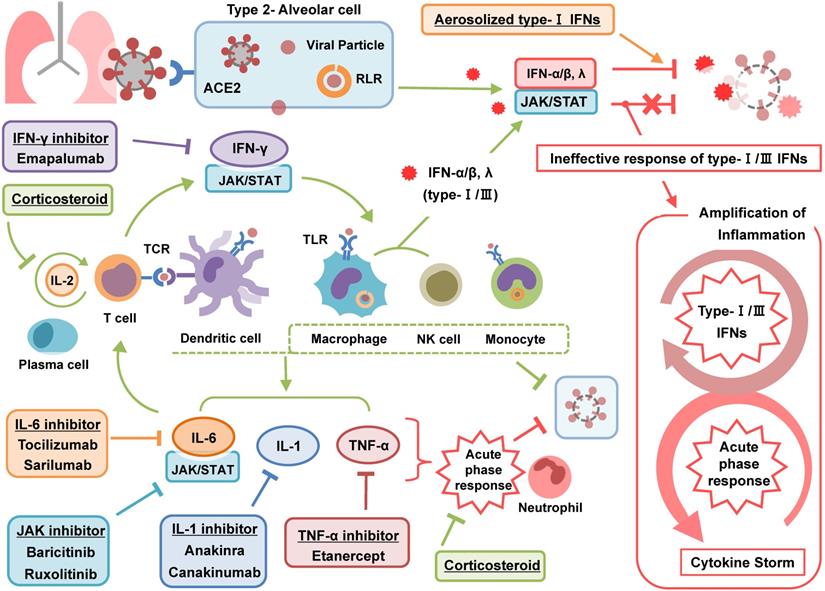
Pathophysiology and treatment of cytokine storm in COVID-19. Type-1 IFNs play a major role in inhibiting the early stage of COVID-19 infection. Dendritic cells and mononuclear macrophages that recognize viral antigens induce an acute phase response through pro-inflammatory cytokines such as IL-6, IL-1, and TNF-α. Among them, IL-6 stimulates T-cells to activate adaptive immunity. Activated T cells also stimulate macrophage and NK cells through IFN-γ to promote virus removal. Failure of the initial immune response by type-1 IFNs increases excessively the activity of immune system leading to cytokine storm. Aerosolized type-1 IFNs promotes an initial immune response to remove virus, corticosteroids and novel cytokine blockades inhibits immune responses to prevent cytokine storm. ACE2: angiotensin converting enzyme-2; COVID-19: coronavirus disease 2019; IFN: interferon; IL: interleukin; JAK/STAT; Janus kinase-signal transducer and activator of transcription; NK: natural killer; RLR: retinoic acid-inducible gene-1-like receptor; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; TCR: T cell receptor; TCZ: tocilizumab; TLR: toll-like receptor; TNF-α: tumor necrosis factor-alpha.
Many researchers believe that there are certain people who are vulnerable to cytokine storm in COVID-19, although clear evidence of this is lacking, at the moment. In support of this notion is a genetic predisposition to cytokine storm in primary HLH, which is already well documented. Primary HLH results from genetic defects in a perforin and granzyme dependent pathway in the action of natural killer (NK) cells and cytotoxic T lymphocytes [31]. Additionally, a few studies have also indicated genetic predispositions in other cytokine storm diseases. One study showed that some patients with SJIA carried perforin gene mutations related to reduce perforin activity [32]. Another study demonstrated that perforin expression was also reduced in patients with SJIA and could be restored following clinical improvement after stem cell transplantation [33]. Accordingly, we suspect there could be a genetic predisposition that makes some people more vulnerable than others to cytokine storm, leading to severe COVID-19 [34, 35]. To confirm this, more epidemiological studies on patients presenting with cytokine storm related to COVID-19 are needed in the future.
Treatment of cytokine storm
Treatments with specific cytokine inhibitors
Current recommendations for the treatment of primary and secondary HLH are based on specific treatment protocols [22]. The mainstay of treatment is the use of corticosteroids, cyclosporine and etoposide, which are commonly used to target dysregulated T cells. The range of effects of these drugs is extensive and inhibits various mechanisms in the pathogenesis of HLH. Specific cytokine blockades have also proven successful in treating various types of HLH, although not as a general therapeutic option: it appears to only be effective when a target cytokine plays a key role in the disease.
During the COVID-19 pandemic, many therapeutic drugs and interventions have been attempted to treat severe COVID-19 (Table 1). In light of successes in the treatment of various types of cytokine storm, a few cytokine blockades have been explored in the treatment of severe COVID-19 [36].
Therapeutic options for the treatment of cytokine storm in severe COVID-19
| Targeted inhibition | Drugs or Interventions | Previous established or alternative indications |
|---|---|---|
| IL-1 | Anakinra, Canakinumab | RA, MAS-HLH |
| IL-6 | Tocilizumab, Sarilumab, Siltuximab | RA, SJIA, CRS, Castleman disease |
| TNF-α | Etanercept | RA, SJIA |
| IFN-γ | Emapalumab | Primary HLH |
| JAK | Baricitinib, Ruxolitinib | RA, MF, PV |
| Non-selective | Glucocorticoid | Various autoimmune diseases and hematologic malignancies |
| Non-selective | Colchicine | Gout |
| Non-selective | Mesenchymal stem cell | Not yet documented, Regenerative medicine |
| Non-selective | Plasma exchange | Various Ab-mediated diseases, Hyperviscosity syndrome |
| Non-selective | Intravenous immunoglobulin | Various autoimmune and infectious diseases, Ab deficiency disorders |
| Non-selective | Convalescent plasma | Not yet documented, Rescue therapy in severe infectious diseases |
| Non-selective | Radiation | Tumor |
Ab: antibody; COVID-19: coronavirus disease 2019; CRS: cytokine release syndrome; HLH: hemophagocytic lymphohistiocytosis; IFN: interferon; IL: interleukin; JAK: Janus kinase; MAS: macrophage activation syndrome; MF: myelofibrosis; PV: polycythemia vera; RA: rheumatoid arthritis; SJIA: systemic juvenile idiopathic arthritis; TNF-α: tumor necrosis factor-alpha.
Inhibition of IL-1 signaling
IL-1 is one of the major pro-inflammatory cytokines. It is comprised of two types of ligands, IL-1α and IL-1ß, of which IL-1ß plays a major role with systemic effects. IL-1 is primarily produced by innate immune cells, such as macrophages and monocytes. IL-1 exerts pro-inflammatory actions to recruit immune cells and to induce secondary cytokine production resulting in acute phase reactions (Figure 1). Some researchers argue that diseases caused by dysregulated IL-1 ought to be considered as auto-inflammatory diseases, not auto-immune diseases, because IL-1 is not directly involved in adaptive immunity, as with T and B cells [37].
Anakinra, a recombinant IL-1 receptor antagonist, has been effectively used to treat auto-inflammatory diseases, from rheumatoid arthritis to cytokine storm [38]. Another IL-1 inhibitor, canakinumab which is a human monoclonal antibody neutralizing IL-1ß, has also been found to be useful in treating various types of rheumatologic diseases, with the advantage of prolonged action [39]. Inhibition of IL-1 naturally exists as a feedback mechanism of self-regulation. Anakinra is a modified form of the human IL-1 inhibitor. In that context, the action of anakinra is expected to be safe because it is similar to its physiologic mechanism. Despite the infection risk, it is remarkably safe, relative to other agents in the same line. Furthermore, the effects of anakinra are not limited to the treatment of rheumatologic diseases. A re-analysis of a randomized controlled trial (RCT) demonstrated that anakinra reduces mortality in sepsis patients experiencing cytokine storm [40].
A few studies have suggested beneficial effects with the use of anakinra in COVID-19. A recent retrospective cohort study of patients with COVID-19 and ARDS showed that high-dose anakinra could be used safely and improved respiratory function [41]. Another prospective cohort study of patients with severe COVID-19 pneumonia demonstrated that anakinra reduced the need for mechanical ventilation and mortality without serious side-effects [42]. Although both studies showed promising results for anakinra on severe COVID-19, further validation is needed through RCTs.
Inhibition of IL-6 signaling
IL-6 is an important pro-inflammatory cytokine that has pleiotropic effects. It is induced by infection or tissue injury and rapidly elicits acute reactions to minimize them. IL-6 promotes the production of various acute phase proteins in hepatocytes and induces the differentiation of immune cells, such as B and T cells (Figure 1). In addition, IL-6 is involved in the metabolism of iron by regulating hepcidin to make a microenvironment prohibitive against infection. Taken together, IL-6 plays a role in priming inflammatory responses and in activating adaptive immunity against infection or injury [24].
A few IL-6 inhibitors have been used in auto-immune disorders and related cytokine storm. Tocilizumab (TCZ), an anti-IL-6 receptor monoclonal antibody, has been used to treat various types of rheumatologic diseases, including rheumatoid arthritis, with excellent efficacy. Furthermore, it has been approved by the FDA in the management of CRS after CAR T-cell therapy [43]. On the other hand, it has not been found to show any effects against sepsis syndrome.
Many studies have demonstrated that IL-6 is significantly elevated in patients with COVID-19. One study showed that IL-6 levels in COVID-19 patients were very high and comparable to those in CAR T cell CRS [44, 45]. Moreover, studies have suggested that IL-6 is predictive of poor outcomes in COVID-19. One study showed that non-survivors had higher IL-6 levels than survivors [46], and another recent study suggested that a high level of IL-6 predicted the risk of requiring mechanical ventilation [47]. A recent prospective cohort study indicated that high levels of IL-6 and d-dimer reflected systemic inflammation and thrombotic condition, and predicted in-hospital mortality of COVID-19 [48].
Two IL-6 inhibitors are being attempted in treatment of COVID-19. In a few case series, sarilumab and tocilizumab have shown beneficial effects in reducing severity and mortality in severe COVID-19. A recent retrospective cohort study of 1,351 patients with COVID-19 and pneumonia showed that tocilizumab significantly reduced the mechanical ventilation risk or death [49]. In an observational study of 154 patients requiring mechanical ventilation, tocilizumab reduced the risk of death by 45%. However, this study also showed that, with tocilizumab, the risk of superinfection significantly increased [50]. Although RCTs are required to validate these clinical effects, the outlook for these drugs in the treatment of COVID-19 seems encouraging.
Inhibition of TNF-α signaling
TNF-α is a cytokine primarily produced by activated macrophages in the acute inflammatory phase (Figure 1). In the TGN1412 trial, TNF-α was first to be elevated among several elevated cytokines [8]. Considering the role of TNF-α in the inflammatory response, TNF-α blockade is expected to have promising effects in various types of cytokine storm. However, in contrast to treatment of rheumatoid arthritis of rheumatoid diseases, its effectiveness has not been proven in the actual treatment of cytokine storm. While a few case reports have described the usefulness of TNF-α blockade with etanercept in MAS [51, 52], there are no relevant studies proving the effects systemically, and two studies failed to document beneficial effects for TNF-α blockade in sepsis syndrome [53, 54].
Anti-TNF-α therapy has not been actively attempted in COVID-19. Reports of COVID-19 infection in patients who maintain anti-TNF-α therapy against existing inflammatory bowel disease have suggested that anti-TNF-α therapy maintenance is safe; however, clear evidence of clinical improvement therewith is lacking [55]. A recent interesting study showed that TNF-α mediated acute lung injury was reduced by using an aptamer targeting TNF-α. Considering that acute lung injury is a characteristic finding in COVID-19, this study provides a possible therapeutic approach to treating COVID-19 cytokine storm accompanied by lung injury [56].
Inhibition of IFN-γ signaling
IFN-γ is secreted by several immune cells, including macrophages, NK cells, and T cells, and plays a role in stimulating the main inflammatory effector cells directly (Figure 1). Thus, IFN-γ is considered a major effector cytokine in various cytokine storm disorders. In particular, IFN-γ is a key cytokine in primary HLH, which is caused by hyperactivity of the immune system in response to a failure to eliminate pathogens due to perforin defects. In HLH, IFN-γ is excessively stimulated and leads to a cytokine storm. Emapalumab, an anti- IFN-γ monoclonal antibody, has been used to treat primary HLH since it was approved by the FDA in 2018 [57]. Several studies have documented clinical effects for emapalumab by confirming decreases in blood levels of C-X-C motif chemokine ligand 9 (CXCL9), an IFN-γ derived chemokine that is a good indicator to measure IFN-γ activity after use of emapalumab. A recent interventional study indicated that emapalumab had clinical effects without specific side effects in primary HLH treatment [58]. Another study described clinical effects for emapalumab in refractory HLH caused by Epstein-Barr virus (EBV) infection [59]. Interestingly, the study indicated that excess IFN-γ contributed to immune paralysis and that emapalumab could reduce cytokine storm and effectively clear persistent EBV by reversing immune paralysis. For treating COVID-19, there are no significant trials supporting the use of IFN-γ inhibitors yet. Instead, many researchers have attempted to administer type 1 IFNs, such as IFN α, ß and κ, in the early phase of COVID-19 [60, 61], as recent studies have suggested that decreased type Ⅰ and Ⅲ IFNs response to SARS-CoV-2 plays an important role in the development of the observed cytokine storm associated with COVID-19 [62].
Inhibition of JAK pathway
JAK is an intracellular tyrosine kinase that mediates signals from cytokines, hormones, and growth factors. The Janus kinase-signal transducer and activator of transcription (JAK/STAT) pathway is commonly involved in various cytokine activation processes (Figure 1). JAK inhibitors, including ruxolitinib and baricitinib, have been used to treat a variety of autoimmune and hematology diseases. JAK inhibition is thought to be able to effectively suppress cytokine storm because it can non-selectively inhibit the activity of many cytokines. On the other hand, non-selective inhibition of the immune response poses a risk of secondary infection, because it also suppresses the innate immune system that fights against pathogenic microorganisms, such as viruses, bacteria, or fungi. Moreover, because the JAK/STAT pathway is involved in several physiological mechanisms other than the immune response, inhibition of these pathways can potentially lead to unexpected side effects.
JAK inhibition in COVID-19 appears to offer two clinical advantages. JAK inhibitors block cytokine signaling, thereby reducing excessive inflammatory responses, as well as the entry of SARS-CoV-2, in the early phase of infection [63]. As is well known, SARS-CoV-2 enters the body through angiotensin converting enzyme-2 (ACE2) on alveolar type 2 cells in the lungs, and several regulators are involved in mediating endocytosis and intracellular transport through ACE2. AP2-associated protein kinase-1, one such regulator, is also a target of JAK inhibitors, particularly baricitinib. Thus, JAK inhibitors can impede the entry and proliferation of SARS-CoV-2 [64]. Accordingly, several JAK inhibitors, including baricitinib, ruxolitinib, and fedratinib, are being studied in the treatment of severe COVID-19. A recent multi-centered RCT of ruxolitinib in patients with COVID-19 noted faster clinical improvement with the drug, although the results lacked statistical significance [65]. Another multi-centered retrospective study demonstrated that baricitinib reduced the rate of ICU admission and fatality and increased discharge rates [66].
Other anti-inflammatory or immunosuppressive agents
In addition to cytokine blockade, various anti-inflammatory therapies are being applied in an attempt to treat COVID-19. This trends show that anti-inflammatory treatment is as important as anti-viral treatment in the treatment of COVID-19. As with any serious illness, many clinicians are currently using glucocorticoids as an empirical treatment for severe COVID-19. Unlike specific cytokine inhibitors, glucocorticoids are believed to be effective against cytokine storm by inhibiting multiple inflammatory targets (Figure 1). Glucocorticoids not only have excellent immunosuppressive effects on immune cells, but also have anti-inflammatory effects by inhibiting the production of major inflammatory molecules, including prostaglandins and leukotrienes. Evidence supporting the use of glucocorticoids in severe COVID-19, however, is lacking [67, 68]. Although this matter is still argued, several studies have positively reported steroid effects in severe COVID-19 [69, 70].
Colchicine is an anti-inflammatory drug that is commonly used in gouty arthritis. The main action of colchicine is to impede the function of neutrophils, and it has the effect of inhibiting IL-1ß activity by inhibition of the inflammasome complex. A few studies are investigating the clinical effects of colchicine on COVID-19 [71]. A recent RCT demonstrated that colchicine delays the time to clinical deterioration in COVID-19 [72].
Some clinicians have attempted to use stem cells for the treatment of severe COVID-19 [73]. Although the effectiveness of stem cell therapy in existing immune diseases has not been clearly demonstrated, researchers have argued that stem cell therapy has immunomodulatory effects and helps to differentiate immune cells.
Interestingly, a few researchers have proposed the use of low-dose radiation therapy to control hyper-inflammatory states in severe COVID-19 [74, 75]. According to the cited studies, low-dose radiation (usually < 1.0 Gy) has immune-modulating effects on immune cells, changing them into an anti-inflammatory phenotype. Thus, in the patients with COVID-19 and ARDS, low-dose radiation on both lungs could potentially facilitate clinical improvements in hyper-inflammatory lung injury.
Several drugs have been experimentally shown to be effective in reducing cytokine storm associated with influenza infection, which is similar to COVID-19. Peroxisome proliferator-activated receptors (PPARs) agonists, cyclooxygenase (COX) inhibitors [76], and sphingosine-1-phosphate receptor 1 (S1P1) [77] agonists have been shown in several experimental studies to be effective in hindering cytokine storm in severe influenza infections. Although S1P1 agonist is known to induce lymphopenia, one study suggested that it might be helpful to mitigate the cytokine storm induced ARDS by preventing trafficking of immune cells to the lungs [78]. A recent review described the use of nanomedicine to modulate macrophage dysfunction in cytokine storm. According to the researches, dysfunctional macrophage is critical in the pathogenesis of cytokine storm, and thus selectively inhibiting dysregulated macrophages using liposomes or synthetic nanoparticles can be a novel therapeutic approach for the treatment of cytokine storm [79]. However, the use of these drugs in these instances is not supported by evidence in human subjects and clinical trials, and requires substantial clinical data.
Issues with treatment of cytokine storm associated with COVID-19
Several studies have shown that cytokine plasma concentrations exhibit diurnal variations. According to a meta-analysis study, IL-6 concentrations are lowest in the morning and peak twice during the time from afternoon to morning [80]. Another study showed that several cytokines, including IL-6 and IL-1, had higher concentrations in the afternoon than in the morning [81]. Both studies commonly proposed diurnal variations in cortisol as a contributing factor to the diurnal variations in the cytokines. It also seems that they may also be affected by different physiological mechanisms that have not yet been identified. Therefore, diurnal variations in these cytokines should be considered in the diagnosis and treatment of cytokine storm and regarded as an important factor when determining the appropriate timing of drug administration in cytokine-targeted therapy.
The cytokine storm caused by COVID-19 appears to be more similar to that in sepsis syndrome than that in HLH in hematologic or rheumatologic diseases. The cytokine storm related to COVID-19 was caused by infection as in the case of sepsis syndrome. As the cytokine storm caused by infections is likely involving more complex mechanisms than that by non-infectious causes, the effect of targeted treatment on a specific cytokine may be limited. Nonetheless, the results of ongoing research do not seem to be discouraging, and additional verification through RCTs is required.
Treatment of COVID-19 is largely divided into two parts: antiviral treatment to inhibit SARS-CoV-2 replication and anti-inflammatory treatment to reduce systemic inflammation. Treatment of the cytokine storm belongs to the latter, and new targeted therapies for cytokines and corticosteroids have been attempted. Anti-inflammatory therapy in severe COVID-19 poses one critical issue. Anti-inflammatory treatment is aimed at clinical improvement through suppression of excessive immune responses; while on the other hand, the immunosuppressive effect of anti-inflammatory treatment paradoxically decreases the clearance of the virus from the body and increases the risk of secondary bacterial infections. In influenza infections, despite no definitive conclusion on their effects and risks, steroids are considered empirical drugs in severe cases. During influenza pandemic periods, many clinicians use steroids to treat serious clinical complications, including ARDS [82]. To compensate for decreased capacity of viral eradication and to avoid the risk of secondary bacterial infections, simultaneous administration of anti-viral agents (e.g., oseltamivir) and broad-spectrum antibiotics are essential in severe influenza infection [83]. On the contrary, the anti-inflammatory treatment for severe COVID-19 is in a very disadvantageous position, because there is no proven effective anti-viral agent at the moment. Although an observational study and a preliminary report from an RCT on the effectiveness of the use of remdesivir in severe COVID-19 have recently been released, further verification through complete and reliable RCTs is required [84, 85]. In COVID-19, immunosuppressive agents, including cytokine blockades and steroids, should be used appropriately only when an excessive immune response is evident, and thus, a proper clinical diagnosis of cytokine storm is essential.
Early detection of cytokine storm and immediate initiation of treatments to reduce severity are essential for the treatment of severe COVID-19. A recent report focused on the rheumatologist's role in COVID-19 era, providing guidelines for the early diagnosis of cytokine storm, and suggested the need for cooperation with rheumatologists as professionals with experience in treating cytokine storm and in using various immunosuppressants [86]. Meanwhile, many studies have emphasized the importance of hyperferritinemia in early diagnosis of cytokine storm [6]. Serum ferritin can be easily measured in clinical laboratories and reflects inflammatory states well. Despite the lack of cut-off values, a significant elevation in ferritin and typical clinical features are suggestive of cytokine storm in COVID-19 [86].
Cytokine removal by blood purification
The efficacies of various immunosuppressive measures on cytokine storm are under investigation. While we are eagerly awaiting such results, other therapeutic measures that are able to reduce cytokine levels within hours of application are readily available. Direct removal of such pro-inflammatory stimuli might reduce the likelihood of progressive organ damage [87], a hallmark of COVID-19.
Many studies have investigated the removal of inflammatory cytokines through blood purification [88]. Continuous renal replacement therapy (CRRT), as a type of hemofiltration or hemodiafiltration method, has shown clinical benefits from removing inflammatory molecules, beyond just replacing impaired kidney function. Previous studies have demonstrated that high doses of hemofiltration afforded better outcomes than standard doses in critical patients. These results suggest that high clearance of inflammatory cytokines might exert clinical benefits [89]. Meanwhile, a systemic review investigating the effects of extracorporeal cytokine removal has indicated that high cut-off techniques remove cytokines more effectively through the larger pores of the membrane [90].
Application of therapeutic plasma exchange in the treatment of cytokine storm
TPE is a more specific method to remove molecules more effectively from plasma than CRRT. It is usually used to treat antibody-mediated severe diseases, such as thrombotic microangiopathies, glomerulonephritis forms, Guillain-Barré syndrome, and others [91]. Although TPE is not accepted as a standard treatment for HLH, a few studies have suggested a promising role for TPE in primary and secondary HLH. One study showed that inflammatory states were reduced after TPE therapy in children with hyperferritinemia and secondary HLH [92], and another case study reported that TPE facilitated a rapid improvement in patients with hemophagocytic syndrome during HLH-2004 protocol based standard therapy [93]. A recent case series showed that early use of TPE along with immunosuppressive therapy was an effective treatment strategy for steroid refractory MAS-HLH [94].
TPE has also been used as an alternative treatment for severe sepsis (Table 2). Nevertheless, there is no clear evidence to recommend the use of TPE in severe sepsis yet, because the available trials have shown conflicting results [95, 96]. Still, a few studies support the role of TPE in sepsis. A well-conducted systemic review indicated that TPE could reduce mortality in adult patients with sepsis [97]. A recent non-randomized prospective study demonstrated that early TPE was associated with a rapid reduction of norepinephrine in septic shock, maintaining mean arterial pressure above 65 mmHg. Furthermore, it could also reduce pro-inflammatory cytokines (IL-6, IL-1ß, and angiopoietin-2) [98]. Another investigation involving subjects with septic shock demonstrated that TPE partially reversed coagulation disorders in patients requiring high doses of norepinephrine [99].
Studies on the effects of therapeutic plasma exchange in severe sepsis or septic shock
| Authors (year) | Type of Study | Subjects | Clinical outcomes |
|---|---|---|---|
| Reeves et al. (1999) | Multicenter, Prospective, RCT | Adults (n, 22); Children (n, 8) with sepsis | No significant difference in mortality at 14-day; No significant reduction in the risk of death in TPE; No effects on IL-6, G-CSF, and thromboxane-B. |
| Busund et al. (2002) | Single-center, Prospective, RCT | Adults (n, 106) with septic shock | Mortality at 28-day: TPE vs. Control; 33·3 vs. 53·8 (%) (p=0·050); Absolute risk reduction: 20·5%. |
| Nguyen et al. (2008) | Single-center, Prospective, RCT | Children (n, 10) with Thrombocytopenia and MOF | Improved ADAMTS-13 activity and organ function; at median 12-day in TPE group (p<0·05). |
| Rimmer et al. (2014) | Systemic review Meta-analysis | Four RCTs including children and adults | No significant reduction of all-cause mortality in overall patients; But, significant reduction of all-cause mortality in adult patients; (Risk Ratio 0·63, CI 0·42-0·96) |
CI: confidence interval; G-CSF: granulocyte colony-stimulating factor; IL: interleukin; JAK: Janus kinase; MOF: multiple organ failure; RCT: randomized controlled trial; TPE: therapeutic plasma exchange.
In regard to TPE use in cytokine storm, TPE has several strengths. First, TPE can elicit immediately clinical effects in rapidly deteriorating cytokine storm. Early use of TPE may compensate for relatively slow action of immunosuppressants. Second, TPE exhibits non-selective properties in the elimination of inflammatory cytokines. It is believed that non-selective removal of cytokines is particularly suitable for the treatment of sepsis-induced cytokine syndrome [100], because the occurrence of a cytokine storm related to sepsis is believed to be the result of a complex mechanism involving various inflammatory factors [101]. In that context, we prefer centrifugal-type TPE over filtration-type, because centrifugal TPE is less selective in separating plasma, although we still believe that TPE itself could provide clinical benefits regardless of the type. A previous study in the patients with Guillain-Barré syndrome also demonstrated that centrifugal TPE had shorter time to onset and greater clinical effects than double filtration plasmapheresis [102]. Last, bulk removal of cytokines by TPE could obviate negative effects on immune cells and improve the function of monocytes and/or macrophages, thereby reversing immune paralysis. Overall, the immunomodulation effects by TPE could reinforce the immunity against COVID-19 and provide the chance of overcoming cytokine storm by increasing susceptibility to simultaneously administered immunosuppressants [100].
Proposal for the use of therapeutic plasma exchange in COVID-19
Influenza infection as a viral disease is more similar to COVID-19 than bacterial sepsis. Severe COVID-19 has clinical features similar to severe influenza infection, characterized by acute lung injury and multiple organ failure. A few case studies have reported that TPE could be helpful in serious influenza infections. One case study showed that three children with severe influenza infections were able to recover from acute respiratory failure and hemodynamic shock through a rescue therapy comprising three consecutive sessions of TPE [103]. Another case study showed that three patients with influenza-associated encephalopathy had markedly improved with two to three sessions of TPE every other day in combination with methylprednisolone injections (30 mg/kg). In addition, the study indicated that a key pro-inflammatory cytokine, IL-6, was significantly reduced in blood and cerebrospinal fluid after repetitive TPE [104].
Currently, remdesivir is being considered as a potential drug for treating COVID-19; however, in severe COVID-19, both antiviral treatment that suppresses the activity of the virus and anti-inflammatory treatment that can reduce excessive inflammation in the body are essential. In our eyes, rescue therapy comprising TPE as an alternative treatment early in serious cases with signs of a rapid worsening disease course and features of cytokine storm might be warranted. Despite the lack of a solid basis for the availability of TPE, even in severe infectious conditions such as sepsis, we believe that practical experiences with TPE use in various clinical diseases provide clues on TPE availability. Furthermore, the simple and reasonable hypothesis that, by exchanging plasma, cytokines and viruses can be effectively eliminated is worth considering. In this regard, a recent case report described the effects of TPE on COVID-19 [105]. In that case report, a patient with severe respiratory failure and anti-phospholipid syndrome by COVID-19 was given three sessions of plasma exchange. After plasma exchange, the patient showed clinical improvements with reduced titers of antiphospholipid antibodies and inflammatory markers, including IL-6. Another case series demonstrated that TPE had effects on treating COVID-19-related autoimmune meningoencephalitis [106]. In that report, ferritin levels reflective of hyper-inflammatory states were reduced significantly after repetitive plasma exchange. Other than that, a few case studies of COVID-19 patients with ARDS and shock have indicated that TPE dramatically improved clinical conditions and inflammatory markers, even after small numbers of trials [107-110]. A recent observational cohort study also showed that TPE afforded clinical benefits of a higher extubation rate and lower mortality rate at 28-day in COVID-19 patients with respiratory failure than non-TPE cases [111]. Currently, while there are increasing case reports indicating the usefulness of TPE use in severe COVID-19 (Table 3) and while many clinicians might be using it in clinical practice, RCTs are still required to attain relevant clinical evidence in support thereof [112].
Case studies on the effects of therapeutic plasma exchange in COVID-19
| Authors | Subjects | Prescription of TPE a | Clinical outcomes |
|---|---|---|---|
| Keith et al. | One patient with pneumonia, shock, and multi-organ failure | 1/ 4.5L/ FFP | Improved respiratory condition and hypotensive shock, increased heart function |
| Shi et al. | One patient with ARDS, shock | 3/ 6L/ FFP | Improved respiratory condition and hypotensive shock |
| Morath et al. | Five patients with respiratory failure | 1~2/ 3.39L/ FFP | Improved respiratory condition and hypotensive shock, Decrease in inflammatory marker (IL6, ferritin, D-dimer) |
| Adeli et al. | Eight patients with ARDS and shock | 3~5/ 2L/ FFP, albumin | Improved respiratory condition |
| Dogan et al. | Six patients with COVID-19-related autoimmune meningoencephalitis | 1~3/ no data/ albumin | Clinical improvement including meningoencephalitis, Decrease in serum ferritin |
| Khamis et al. | Eleven patients with ARDS or pneumonia | 5/ one times body plasma volume/ FFP | Higher extubation rate, lower mortality at 14 and 28 days compared to non-TPE cases |
a Prescription of TPE is presented as total number of trials/ dose per session/ type of substitution fluid. ARDS: acute respiratory distress syndrome; COVID-19: coronavirus disease 2019; FFP: fresh frozen plasma; IL: interleukin; TPE: therapeutic plasma exchange.
There are several clinical issues with the use of TPE in COVID-19. First, it poses risks of bleeding and catheter infection. In addition, it can cause electrolyte imbalances, such as hypocalcemia and hypokalemia, depending on the type of anti-coagulating agents or replacement fluids. TPE can also occasionally induce unpredicted anaphylactic shock due to the use of blood materials. However, the incidences of these complications are thought to be relatively small, and most cases can be controlled with medical support. Although TPE can be applied without technical difficulties in experienced centers, active involvement of medical staff in this procedure and capacity issues (most centers are limited with machines) only allow for the use of TPE in selected patients. Above all, the primary critical issue with TPE in COVID-19 is that the use of non-convalescent plasma as a substitution fluid could reduce protective antibodies against SARS-CoV-2. According to the clinical experiences of a few researchers, SARS-CoV-2-specific IgG and IgA antibodies were detected in waste bags, and circulating antibodies were also reduced during TPE application [113]. However, despite these concerns, a few studies have demonstrated the clinical benefits of TPE in severe COVID-19, and we believe that timely, appropriate application of TPE in selected cases presenting with cytokine storm could be beneficial.
Since clinical deterioration of COVID-19 is mostly accompanied by worsening pneumonia and development of ARDS, we recommend initiating TPE when there are signs of respiratory failure requiring mechanical ventilation. In particular, highly elevated ferritin levels in blood, and the estimation of myocardial injury using high-sensitivity cardiac troponin I may also be considered useful [114, 115].
Severe COVID-19 is usually accompanied by hypotensive shock, which makes it difficult to maintain extracorporeal circulation. Therefore, it is better to start TPE early when blood pressure is maintained. In addition, referring to existing reports, we believe that TPE may be appropriate for use every day, or every other day. Previous reports have generally applied TPE two to three times in total; however, we believe that TPE can continue as long as there is no particular complication and exchangeable plasma is supplied smoothly. In addition, discontinuation may be considered if critical clinical conditions can be controlled by therapeutic methods other than TPE. In a few studies, two to 14 daily sessions, on average, were performed, with close monitoring of disseminated intravascular coagulation markers [116, 117]. A target calculated plasma volume exceeding 1 should be considered, and fresh frozen plasma is recommended to be used as a substitution fluid, due to the replenishment of anti-infective factors in serum. Ideally, convalescent plasma, if available, should be used, even if only at the end of a session [118-120]. Regarding existing TPE techniques, the centrifugal method might be preferable, as RCTs on TPE in septic shock have showed positive results with using centrifugation, although filtration types are also considered useful. Also, precise fluid overload control is necessary as decreasing vessel permeability is positive but can further impair pulmonary saturation.
Conclusion
Cytokine storm is a severe clinical condition caused by systemic hyper-inflammation, resulting in severe injury to multiple organs, including the lungs, and even death. In that sense, anti-inflammatory treatment, along with anti-viral therapy, remains an important part of treatment for COVID-19. Currently, many anti-inflammatory agents have been attempted to treat severe COVID-19, and a large number of mixed results are being reported. With mounting evidence, we believe novel cytokine inhibitors, including tocilizumab, anakinra, and baricitinib, are promising options for treating severe COVID-19.
In this review, we described TPE as a realistic alternative in the treatment of cytokine storm associated with COVID-19. We believe that TPE can have clinical benefits on severe COVID-19 when initiated promptly after early diagnosis based on rapid clinical deterioration and high inflammatory parameters, such as serum ferritin and high-sensitivity cardiac troponin I. We strongly recommend using TPE with convalescent plasma. Overall, we propose that TPE can be useful in combination with other potentially effective options for treating severe COVID-19.
Abbreviations
Ab: antibody; ACE2: angiotensin converting enzyme 2; ARDS: acute respiratory distress syndrome; CAR: chimeric antigen receptor; COVID-19: coronavirus disease 2019; COX: cyclooxygenase; CRRT: continuous renal replacement therapy; CRS: cytokine release syndrome; CXCL9: C-X-C motif chemokine ligand 9; DAMP: damage-associated molecular pattern; EBV: Epstein-Barr virus; FFP: fresh frozen plasma; G-CSF: granulocyte colony-stimulating factor; HLH: hemophagocytic lymphohistiocytosis; ICU: intensive care unit; IFN: interferon; IL: interleukin; IP10: IFN-γ-induced protein 10; JAK: Janus kinase; JAK/STAT: Janus kinase/signal transducers and activators of transcription; MAS: macrophage activation syndrome; MCP1: monocyte chemoattractant protein 1; MF: myelofibrosis; MOF: multiple organ failure; NK: natural killer; PAMP: pathogen-associated molecular pattern; PPARs: Peroxisome proliferator-activated receptors; PRRs: pattern recognition receptors; PV: polycythemia vera; RA: rheumatoid arthritis; RCT: randomized controlled trial; RLR: retinoic acid-inducible gene-1-like receptor; S1P1: sphingosine-1-phosphate receptor 1; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; SJIA: systemic juvenile idiopathic arthritis; TCR: T cell receptor; TCZ: tocilizumab; TLR: toll-like receptor; TNF-α: tumor necrosis factor-alpha; TPE: therapeutic plasma exchange.
Contributions
All authors made substantial contributions to all of the following: (1) conception and design of the study, data acquisition, or analysis and interpretation of data; (2) drafting or critical revision of the article for intellectual content; and (3) approval of the final version of the submitted manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun. 2020 p:102433
2. Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H. et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8(5):475-81
3. Behrens EM, Koretzky GA. Review: Cytokine Storm Syndrome: Looking Toward the Precision Medicine Era. Arthritis Rheumatol. 2017;69(6):1135-43
4. Ferrara JL, Abhyankar S, Gilliland DG. Cytokine storm of graft-versus-host disease: a critical effector role for interleukin-1. Transplant Proc. 1993;25(1 Pt 2):1216-7
5. Favalli EG, Ingegnoli F, De Lucia O, Cincinelli G, Cimaz R, Caporali R. COVID-19 infection and rheumatoid arthritis: Faraway, so close!. Autoimmun Rev. 2020 p:102523
6. Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033-4
7. Stegmayr BG. Plasmapheresis in severe sepsis or septic shock. Blood Purif. 1996;14(1):94-101
8. Suntharalingam G, Perry MR, Ward S, Brett SJ, Castello-Cortes A, Brunner MD. et al. Cytokine storm in a phase 1 trial of the anti-CD28 monoclonal antibody TGN1412. N Engl J Med. 2006;355(10):1018-28
9. Amaral MC, Alves JD. Pathogenesis of multi-organic failure in autoimmune diseases. Autoimmun Rev. 2009;8(6):525-8
10. Henter JI, Elinder G, Soder O, Hansson M, Andersson B, Andersson U. Hypercytokinemia in familial hemophagocytic lymphohistiocytosis. Blood. 1991;78(11):2918-22
11. Esteban YM, de Jong JLO, Tesher MS. An Overview of Hemophagocytic Lymphohistiocytosis. Pediatr Ann. 2017;46(8):e309-e13 doi: 10.3928/19382359-20170717-01
12. Carter SJ, Tattersall RS, Ramanan AV. Macrophage activation syndrome in adults: recent advances in pathophysiology, diagnosis and treatment. Rheumatology (Oxford). 2019;58(1):5-17
13. Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR. et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013;368(16):1509-18
14. Machowicz R, Janka G, Wiktor-Jedrzejczak W. Similar but not the same: Differential diagnosis of HLH and sepsis. Crit Rev Oncol Hematol. 2017;114:1-12
15. Jordan MB, Hildeman D, Kappler J, Marrack P. An animal model of hemophagocytic lymphohistiocytosis (HLH): CD8+ T cells and interferone gamma are essential for the disorder. Blood. 2004;104(3):735-43
16. Pascual V, Allantaz F, Arce E, Punaro M, Banchereau J. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J Exp Med. 2005;201(9):1479-86
17. Mazodier K, Marin V, Novick D, Farnarier C, Robitail S, Schleinitz N. et al. Severe imbalance of IL-18/IL-18BP in patients with secondary hemophagocytic syndrome. Blood. 2005;106(10):3483-9
18. Barrett DM, Teachey DT, Grupp SA. Toxicity management for patients receiving novel T-cell engaging therapies. Curr Opin Pediatr. 2014;26(1):43-9
19. Muller N, van den Brandt J, Odoardi F, Tischner D, Herath J, Flugel A. et al. A CD28 superagonistic antibody elicits 2 functionally distinct waves of T cell activation in rats. J Clin Invest. 2008;118(4):1405-16
20. St Clair EW. The calm after the cytokine storm: lessons from the TGN1412 trial. J Clin Invest. 2008;118(4):1344-7
21. Hayden A, Lin M, Park S, Pudek M, Schneider M, Jordan MB. et al. Soluble interleukin-2 receptor is a sensitive diagnostic test in adult HLH. Blood Adv. 2017;1(26):2529-34
22. La Rosee P, Horne A, Hines M, von Bahr Greenwood T, Machowicz R, Berliner N. et al. Recommendations for the management of hemophagocytic lymphohistiocytosis in adults. Blood. 2019;133(23):2465-77
23. Punt J, Stranford S, Jones P, Owen J. Kuby Immunology. 8th ed. US: Freeman WH & Co publisher. 2018
24. Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. 2014;6(10):a016295
25. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497-506
26. Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846-8
27. McGonagle D, Sharif K, O'Regan A, Bridgewood C. The Role of Cytokines including Interleukin-6 in COVID-19 induced Pneumonia and Macrophage Activation Syndrome-Like Disease. Autoimmun Rev. 2020 p:102537
28. Ye Q, Wang B, Mao J. The pathogenesis and treatment of the `Cytokine Storm' in COVID-19. The Journal of infection. 2020;80(6):607-13
29. Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Moller R. et al. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell. 2020;181(5):1036-45.e9
30. Hadjadj J, Yatim N, Barnabei L, Corneau A, Boussier J, Smith N. et al. Impaired type I interferone activity and inflammatory responses in severe Covid-19 patients. Science. 2020;369(6504):718-24
31. Jenkins MR, Rudd-Schmidt JA, Lopez JA, Ramsbottom KM, Mannering SI, Andrews DM. et al. Failed CTL/NK cell killing and cytokine hypersecretion are directly linked through prolonged synapse time. J Exp Med. 2015;212(3):307-17
32. Vastert SJ, van Wijk R, D'Urbano LE, de Vooght KM, de Jager W, Ravelli A. et al. Mutations in the perforin gene can be linked to macrophage activation syndrome in patients with systemic onset juvenile idiopathic arthritis. Rheumatology (Oxford). 2010;49(3):441-9
33. Wulffraat NM, Rijkers GT, Elst E, Brooimans R, Kuis W. Reduced perforin expression in systemic juvenile idiopathic arthritis is restored by autologous stem-cell transplantation. Rheumatology (Oxford). 2003;42(2):375-9
34. Trouillet-Assant S, Viel S, Gaymard A, Pons S, Richard JC, Perret M. et al. Type I IFN immunoprofiling in COVID-19 patients. J Allergy Clin Immunol. 2020;146(1):206-8.e2
35. Initiative C-HG. The COVID-19 Host Genetics Initiative, a global initiative to elucidate the role of host genetic factors in susceptibility and severity of the SARS-CoV-2 virus pandemic. Eur J Hum Genet. 2020;28(6):715-8
36. Zhang W, Zhao Y, Zhang F, Wang Q, Li T, Liu Z. et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clin Immunol. 2020;214:108393
37. Doria A, Zen M, Bettio S, Gatto M, Bassi N, Nalotto L. et al. Autoinflammation and autoimmunity: bridging the divide. Autoimmun Rev. 2012;12(1):22-30
38. Miettunen PM, Narendran A, Jayanthan A, Behrens EM, Cron RQ. Successful treatment of severe paediatric rheumatic disease-associated macrophage activation syndrome with interleukin-1 inhibition following conventional immunosuppressive therapy: case series with 12 patients. Rheumatology (Oxford). 2011;50(2):417-9
39. Cavalli G, Dinarello CA. Anakinra Therapy for Non-cancer Inflammatory Diseases. Front Pharmacol. 2018;9:1157
40. Shakoory B, Carcillo JA, Chatham WW, Amdur RL, Zhao H, Dinarello CA. et al. Interleukin-1 Receptor Blockade Is Associated With Reduced Mortality in Sepsis Patients With Features of Macrophage Activation Syndrome: Reanalysis of a Prior Phase III Trial. Crit Care Med. 2016;44(2):275-81
41. Cavalli G, De Luca G, Campochiaro C, Della-Torre E, Ripa M, Canetti D. et al. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study. Lancet Rheumatol. 2020;2(6):e325-e31 doi: 10.1016/S2665-9913(20)30127-2
42. Huet T, Beaussier H, Voisin O, Jouveshomme S, Dauriat G, Lazareth I. et al. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol. 2020 e393-e400. doi: 10.1016/S2665-9913(20)30164-8
43. Lee DW, Gardner R, Porter DL, Louis CU, Ahmed N, Jensen M. et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124(2):188-95
44. Hoiland RL, Stukas S, Cooper J, Thiara S, Chen LYC, Biggs CM. et al. Amelioration of COVID-19 related cytokine storm syndrome: Parallels to chimeric antigen receptor-T cell cytokine release syndrome. Br J Haematol. 2020;190(3):e150-e4 doi: 10.1111/bjh.16961
45. England JT, Abdulla A, Biggs CM, Lee AYY, Hay KA, Hoiland RL. et al. Weathering the COVID-19 storm: Lessons from hematologic cytokine syndromes. Blood Rev. 2020 p:100707
46. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-62
47. Herold T, Jurinovic V, Arnreich C, Lipworth BJ, Hellmuth JC, Bergwelt-Baildon MV. et al. Elevated levels of IL-6 and CRP predict the need for mechanical ventilation in COVID-19. J Allergy Clin Immunol. 2020;146(1):128-36.e4
48. Cummings MJ, Baldwin MR, Abrams D, Jacobson SD, Meyer BJ, Balough EM. et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020;395(10239):1763-70
49. Campochiaro C, Della-Torre E, Cavalli G, De Luca G, Ripa M, Boffini N. et al. Efficacy and safety of tocilizumab in severe COVID-19 patients: a single-centre retrospective cohort study. Eur J Intern Med. 2020;76:43-9
50. Somers E, Eschenauer G, Troost J, Golob J, Gandhi T, Wang L. et al. Tocilizumab for treatment of mechanically ventilated patients with COVID-19. Clin Infect Dis. 2020 ciaa954. doi: 10.1093/cid/ciaa954
51. Makay B, Yilmaz S, Turkyilmaz Z, Unal N, Oren H, Unsal E. Etanercept for therapy-resistant macrophage activation syndrome. Pediatr Blood Cancer. 2008;50(2):419-21
52. Maeshima K, Ishii K, Iwakura M, Akamine M, Hamasaki H, Abe I. et al. Adult-onset Still's disease with macrophage activation syndrome successfully treated with a combination of methotrexate and etanercept. Mod Rheumatol. 2012;22(1):137-41
53. Abraham E, Wunderink R, Silverman H, Perl TM, Nasraway S, Levy H. et al. Efficacy and safety of monoclonal antibody to human tumor necrosis factor alpha in patients with sepsis syndrome. A randomized, controlled, double-blind, multicenter clinical trial. TNF-alpha MAb Sepsis Study Group. JAMA. 1995;273(12):934-41
54. Clark MA, Plank LD, Connolly AB, Streat SJ, Hill AA, Gupta R. et al. Effect of a chimeric antibody to tumor necrosis factor-alpha on cytokine and physiologic responses in patients with severe sepsis-a randomized, clinical trial. Crit Care Med. 1998;26(10):1650-9
55. Feldmann M, Maini RN, Woody JN, Holgate ST, Winter G, Rowland M. et al. Trials of anti-tumour necrosis factor therapy for COVID-19 are urgently needed. Lancet. 2020;395(10234):1407-9
56. Lai W, Wang J, Huang B, Lin EP, Yang P. A novel TNF-α-targeting aptamer for TNF-α-mediated acute lung injury and acute liver failure. Theranostics. 2019;9(6):1741-51
57. Vallurupalli M, Berliner N. Emapalumab for the treatment of relapsed/refractory hemophagocytic lymphohistiocytosis. Blood. 2019;134(21):1783-6
58. Locatelli F, Jordan MB, Allen C, Cesaro S, Rizzari C, Rao A. et al. Emapalumab in Children with Primary Hemophagocytic Lymphohistiocytosis. N Engl J Med. 2020;382(19):1811-22
59. Lounder DT, Bin Q, de Min C, Jordan MB. Treatment of refractory hemophagocytic lymphohistiocytosis with emapalumab despite severe concurrent infections. Blood Adv. 2019;3(1):47-50
60. Zhou Q, Chen V, Shannon CP, Wei X, Xiang X, Wang X. et al. Interferon-a2b treatment for COVID-19. Front Immunol. 2020;11:1061
61. Meng Z, Wang T, Chen L, Chen X, Li L, Qin X. et al. An experimental trial of recombinant human interferon alpha nasal drops to prevent COVID-19 in medical staff in an epidemic area. medRxiv. 2020 04.11,20061473
62. Acharya D, Liu G, Gack MU. Dysregulation of type I interferone responses in COVID-19. Nat Rev Immunol. 2020;20(7):397-8
63. Seif F, Aazami H, Khoshmirsafa M, Kamali M, Mohsenzadegan M, Pornour M. et al. JAK Inhibition as a New Treatment Strategy for Patients with COVID-19. Int Arch Allergy Immunol. 2020;181(6):467-75
64. Richardson P, Griffin I, Tucker C, Smith D, Oechsle O, Phelan A. et al. Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. Lancet. 2020;395(10223 e):30-31 doi:10.1016/S0140-6736(20)30304-4
65. Cao Y, Wei J, Zou L, Jiang T, Wang G, Chen L. et al. Ruxolitinib in treatment of severe coronavirus disease 2019 (COVID-19): A multicenter, single-blind, randomized controlled trial. J Allergy Clin Immunol. 2020;146(1):137-46.e3
66. Cantini F, Niccoli L, Nannini C, Matarrese D, Natale MED, Lotti P. et al. Beneficial impact of Baricitinib in COVID-19 moderate pneumonia; multicentre study. The Journal of infection. 2020; S0163-4453(20)30433-3. doi: 10.1016/j.jinf. 2020 06.052
67. Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395(10223):473-5
68. Li H, Chen C, Hu F, Wang J, Zhao Q, Gale RP. et al. Impact of corticosteroid therapy on outcomes of persons with SARS-CoV-2, SARS-CoV, or MERS-CoV infection: a systematic review and meta-analysis. Leukemia. 2020;34(6):1503-11
69. Shang L, Zhao J, Hu Y, Du R, Cao B. On the use of corticosteroids for 2019-nCoV pneumonia. Lancet. 2020;395(10225):683-4
70. Lee KH, Yoon S, Jeong GH, Kim JY, Han YJ, Hong SH. et al. Efficacy of corticosteroids in patients with SARS, MERS and COVID-19: a systematic review and meta-analysis. J Clin Med. 2020;9(8):2392
71. Deftereos S, Giannopoulos G, Vrachatis DA, Siasos G, Giotaki SG, Cleman M. et al. Colchicine as a potent anti-inflammatory treatment in COVID-19: can we teach an old dog new tricks? Eur Heart J Cardiovasc Pharmacother. 2020;6(4):255
72. Deftereos SG, Giannopoulos G, Vrachatis DA, Siasos GD, Giotaki SG, Gargalianos P. et al. Effect of Colchicine vs Standard Care on Cardiac and Inflammatory Biomarkers and Clinical Outcomes in Patients Hospitalized With Coronavirus Disease 2019: The GRECCO-19 Randomized Clinical Trial. JAMA Netw Open. 2020;3(6):e2013136. doi: 10.1001/jamanetworkopen.2020.13136
73. Leng Z, Zhu R, Hou W, Feng Y, Yang Y, Han Q. et al. Transplantation of ACE2(-) Mesenchymal Stem Cells Improves the Outcome of Patients with COVID-19 Pneumonia. Aging Dis. 2020;11(2):216-28
74. Dhawan G, Kapoor R, Dhawan R, Singh R, Monga B, Giordano J. et al. Low dose radiation therapy as a potential life-saving treatment for COVID-19-induced acute respiratory distress syndrome (ARDS). Radiother Oncol. 2020;147:212-6
75. Cosset JM, Deutsch É, Bazire L, Mazeron J-J, Chargari C. Low dose lung radiotherapy for COVID-19-related cytokine storm syndrome: Why not? Cancer Radiother. 2020;24(3):179-81
76. Zheng BJ, Chan KW, Lin YP, Zhao GY, Chan C, Zhang HJ. et al. Delayed antiviral plus immunomodulator treatment still reduces mortality in mice infected by high inoculum of influenza A/H5N1 virus. Proc Natl Acad Sci U S A. 2008;105(23):8091-6
77. Marsolais D, Hahm B, Walsh KB, Edelmann KH, McGavern D, Hatta Y. et al. A critical role for the sphingosine analog AAL-R in dampening the cytokine response during influenza virus infection. Proc Natl Acad Sci U S A. 2009;106(5):1560-5
78. Oldstone MBA, Rosen H. Cytokine storm plays a direct role in the morbidity and mortality from influenza virus infection and is chemically treatable with a single sphingosine-1-phosphate agonist molecule. Curr Top Microbiol Immunol. 2014;378:129-47
79. Liu J, Wan M, Lyon CJ, Hu TY. Nanomedicine therapies modulating macrophage dysfunction: a potential strategy to attenuate cytokine storms in severe infections. Theranostics. 2020;10(21):9591-600
80. Nilsonne G, Lekander M, Åkerstedt T, Axelsson J, Ingre M, Diurnal variation of circulating interleukin-6 in humans. a meta-analysis. PLoS One. 2016;11(11):e0165799. doi: 10.1371/journal.pone.0165799
81. Altara R, Manca M, Hermans KCM, Daskalopoulos EP, Rocca HBPL, Hermans RJJ. Diurnal rhythms of serum and plasma cytokine profiles in healthy elderly individuals assessed using membrane based multiplexed immunoassay. J Transl Med. 2015;13:129
82. Liu Q, Zhou YH, Yang ZQ. The cytokine storm of severe influenza and development of immunomodulatory therapy. Cell Mol Immunol. 2016;13(1):3-10
83. Wong JP, Viswanathan S, Wang M, Sun LQ, Clark GC, D'Elia RV. Current and future developments in the treatment of virus-induced hypercytokinemia. Future Med Chem. 2017;9(2):169-78
84. Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A. et al. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med. 2020;382(24):2327-2336
85. Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, Remdesivir for the treatment of Covid-19 - preliminary report. N Engl J Med. 2020;NEJMoa2007764. doi: 10.1056/NEJMoa2007764.
86. Cron RQ, Chatham WW. The Rheumatologist's Role in Covid-19. J Rheumatol. 2020;47(5):639-42
87. Yang SL, Xu XJ, Tang YM, Song H, Xu WQ, Zhao FY. et al. Associations between inflammatory cytokines and organ damage in pediatric patients with hemophagocytic lymphohistiocytosis. Cytokine. 2016;85:14-7
88. Honore PM, Hoste E, Molnar Z, Jacobs R, Joannes-Boyau O, Malbrain M. et al. Cytokine removal in human septic shock: Where are we and where are we going? Ann Intensive Care. 2019;9(1):56
89. Ronco C, Bellomo R, Homel P, Brendolan A, Dan M, Piccinni P. et al. Effects of different doses in continuous veno-venous haemofiltration on outcomes of acute renal failure: a prospective randomised trial. Lancet. 2000;356(9223):26-30
90. Atan R, Crosbie DC, Bellomo R. Techniques of extracorporeal cytokine removal: a systematic review of human studies. Ren Fail. 2013;35(8):1061-70
91. Clark WF, Huang SS, Walsh MW, Farah M, Hildebrand AM, Sontrop JM. Plasmapheresis for the treatment of kidney diseases. Kidney international. 2016;90(5):974-84
92. Demirkol D, Yildizdas D, Bayrakci B, Karapinar B, Kendirli T, Koroglu TF. et al. Hyperferritinemia in the critically ill child with secondary hemophagocytic lymphohistiocytosis/sepsis/multiple organ dysfunction syndrome/macrophage activation syndrome: what is the treatment? Crit Care. 2012;16(2):R52
93. Bosnak M, Erdogan S, Aktekin EH, Bay A. Therapeutic plasma exchange in primary hemophagocytic lymphohistiocytosis: Reports of two cases and a review of the literature. Transfus Apher Sci. 2016;55(3):353-6
94. Lorenz G, Schul L, Schraml F, Riedhammer KM, Einwachter H, Verbeek M. et al. Adult macrophage activation syndrome-haemophagocytic lymphohistiocytosis: 'of plasma exchange and immunosuppressive escalation strategies' - a single centre reflection. Lupus. 2020;29(3):324-33
95. Reeves JH, Butt WW, Shann F, Layton JE, Stewart A, Waring PM. et al. Continuous plasmafiltration in sepsis syndrome. Plasmafiltration in Sepsis Study Group. Crit Care Med. 1999;27(10):2096-104
96. Busund R, Koukline V, Utrobin U, Nedashkovsky E. Plasmapheresis in severe sepsis and septic shock: a prospective, randomised, controlled trial. Intensive Care Med. 2002;28(10):1434-9
97. Rimmer E, Houston BL, Kumar A, Abou-Setta AM, Friesen C, Marshall JC. et al. The efficacy and safety of plasma exchange in patients with sepsis and septic shock: a systematic review and meta-analysis. Crit Care. 2014;18(6):699
98. Knaup H, Stahl K, Schmidt BMW, Idowu TO, Busch M, Wiesner O. et al. Early therapeutic plasma exchange in septic shock: a prospective open-label nonrandomized pilot study focusing on safety, hemodynamics, vascular barrier function, and biologic markers. Crit Care. 2018;22(1):285
99. Stahl K, Schmidt JJ, Seeliger B, Schmidt BMW, Welte T, Haller H. et al. Effect of therapeutic plasma exchange on endothelial activation and coagulation-related parameters in septic shock. Crit Care. 2020;24(1):71
100. Reeves HM, Winters JL. The mechanisms of action of plasma exchange. Br J Haematol. 2014;164(3):342-51
101. Schwartz J, Winters JL, Padmanabhan A, Balogun RA, Delaney M, Linenberger ML. et al. Guidelines on the use of therapeutic apheresis in clinical practice-evidence-based approach from the Writing Committee of the American Society for Apheresis: the sixth special issue. J Clin Apher. 2013;28(3):145-284
102. Lyu RK, Chen WH, Hsieh ST. Plasma exchange versus double filtration plasmapheresis in the treatment of Guillain-Barre syndrome. Ther Apher. 2002;6(2):163-6
103. Patel P, Nandwani V, Vanchiere J, Conrad SA, Scott LK. Use of therapeutic plasma exchange as a rescue therapy in 2009 pH1N1 influenza A-an associated respiratory failure and hemodynamic shock. Pediatr Crit Care Med. 2011;12(2):e87-9 doi: 10.1097/PCC.0b013e3181e2a569
104. Kawashima H, Togashi T, Yamanaka G, Nakajima M, Nagai M, Aritaki K. et al. Efficacy of plasma exchange and methylprednisolone pulse therapy on influenza-associated encephalopathy. The Journal of infection. 2005;51(2):E53-6 doi: 10.1016/j.jinf.2004.08.017
105. Ma J, Xia P, Zhou Y, Liu Z, Zhou X, Wang J. et al. Potential effect of blood purification therapy in reducing cytokine storm as a late complication of critically ill COVID-19. Clin Immunol. 2020;214:108408
106. Dogan L, Kaya D, Sarikaya T, Zengin R, Dincer A, Akinci IO. et al. Plasmapheresis treatment in COVID-19-related autoimmune meningoencephalitis: Case series. Brain Behav Immun. 2020;87:155-8
107. Keith P, Day M, Choe C, Perkins L, Moyer L, Hays E. et al. The successful use of therapeutic plasma exchange for severe COVID-19 acute respiratory distress syndrome with multiple organ failure. SAGE Open Med Case Rep. 2020;8:2050313X20933473. doi: 10.1177/2050313X20933473
108. Morath C, Weigand MA, Zeier M, Speer C, Tiwari-Heckler S, Merle U. et al. Plasma exchange in critically ill COVID-19 patients. Crit Care. 2020;24(1):481
109. Adeli SH, Asghari A, Tabarraii R, Shajari R, Afshari S, Kalhor N. et al. Therapeutic plasma exchange as a rescue therapy in patients with coronavirus disease 2019: a case series. Pol Arch Intern Med. 2020;130(5):455-458
110. Shi H, Zhou C, He P, Huang S, Duan Y, Wang X. et al. Successful treatment with plasma exchange followed by intravenous immunoglobulin in a critically ill patient with COVID-19. Int J Antimicrob Agents. 2020;56(2):105974
111. Khamis F, Al-Zakwani I, Hashmi SA, Dowaiki SA, Bahrani MA, Pandak N. et al. Therapeutic plasma exchange in adults with severe COVID-19 infection. Int J Infect Dis. 2020;99:214-8
112. Faqihi F, Alharthy A, Alodat M, Asad D, Aletreby W, Kutsogiannis DJ. et al. A pilot study of therapeutic plasma exchange for serious SARS CoV-2 disease (COVID-19): A structured summary of a randomized controlled trial study protocol. Trials. 2020;21(1):506
113. Stahl K, Bode C, David S. First do no harm-beware the risk of therapeutic plasma exchange in severe COVID-19. Crit Care. 2020;24(1):363
114. Cao J, Zheng Y, Luo Z, Mei Z, Yao Y, Liu Z. et al. Myocardial injury and COVID-19: serum hs-cTnI level in risk stratification and the prediction of 30-day fatality in COVID-19 patients with no prior cardiovascular disease. Theranostics. 2020;10(21):9663-73
115. Tersalvi G, Veronese G, Winterton D. Emerging evidence of myocardial injury in COVID-19: a path through the smoke. Theranostics. 2020;10(21):9888-9
116. Nguyen TC, Han YY, Kiss JE, Hall MW, Hassett AC, Jaffe R. et al. Intensive plasma exchange increases a disintegrin and metalloprotease with thrombospondin motifs-13 activity and reverses organ dysfunction in children with thrombocytopenia-associated multiple organ failure. Crit Care Med. 2008;36(10):2878-87
117. Stegmayr BG, Banga R, Berggren L, Norda R, Rydvall A, Vikerfors T. Plasma exchange as rescue therapy in multiple organ failure including acute renal failure. Crit Care Med. 2003;31(6):1730-6
118. Chen L, Xiong J, Bao L, Shi Y. Convalescent plasma as a potential therapy for COVID-19. Lancet Infect Dis. 2020;20(4):398-400
119. Shen C, Wang Z, Zhao F, Yang Y, Li J, Yuan J. et al. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA. 2020
120. Roback JD, Guarner J. Convalescent Plasma to Treat COVID-19: Possibilities and Challenges. JAMA. 2020;323(16):1561-2
Author contact
Corresponding author: Prof. Jae Il Shin, MD, PhD, Address: Yonsei-ro 50, Seodaemun-gu, C.P.O. Box 8044, Department of Pediatrics, Yonsei University College of Medicine, Seoul 03722, Korea. Tel.: +82-2-2228-2050; Fax: +82-2-393-9118; E-mail: shinjiac.
 Global reach, higher impact
Global reach, higher impact