Impact Factor
Theranostics 2021; 11(1):410-425. doi:10.7150/thno.51046 This issue Cite
Research Paper
In vivo multimodal imaging of adenosine A1 receptors in neuroinflammation after experimental stroke
1. Achucarro Basque Center for Neuroscience, 48940 Leioa, Spain.
2. CIC biomaGUNE, Basque Research and Technology Alliance (BRTA), Paseo Miramon 182, 20014, San Sebastian, Spain.
3. Department of Neuroscience, University of Basque Country (UPV/EHU) and CIBERNED, 48940 Leioa, Spain.
4. Departamento de Química Física, Facultad de Ciencia y Tecnología, Universidad del País Vasco (UPV/EHU), 48940, Leioa, Spain.
5. Ikerbasque Basque Foundation for Science, 48013 Bilbao, Spain.
6. National Institute of Radiological Sciences, National Institutes for Quantum and Radiological Science and Technology, Chiba, Japan.
7. Centro de Investigación Biomédica en Red - Enfermedades Respiratorias, CIBERES, 28029 Madrid, Spain.
Abstract
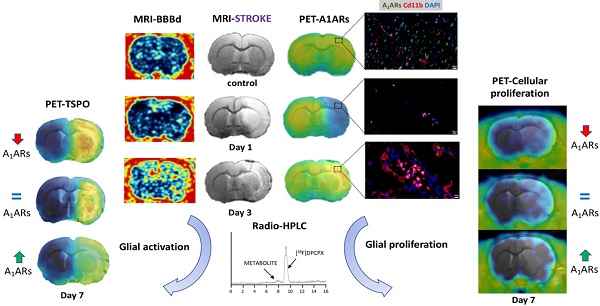
Adenosine A1 receptors (A1ARs) are promising imaging biomarkers and targets for the treatment of stroke. Nevertheless, the role of A1ARs on ischemic damage and its subsequent neuroinflammatory response has been scarcely explored so far.
Methods: In this study, the expression of A1ARs after transient middle cerebral artery occlusion (MCAO) was evaluated by positron emission tomography (PET) with [18F]CPFPX and immunohistochemistry (IHC). In addition, the role of A1ARs on stroke inflammation using pharmacological modulation was assessed with magnetic resonance imaging (MRI), PET imaging with [18F]DPA-714 (TSPO) and [18F]FLT (cellular proliferation), as well as IHC and neurofunctional studies.
Results: In the ischemic territory, [18F]CPFPX signal and IHC showed the overexpression of A1ARs in microglia and infiltrated leukocytes after cerebral ischemia. Ischemic rats treated with the A1AR agonist ENBA showed a significant decrease in both [18F]DPA-714 and [18F]FLT signal intensities at day 7 after cerebral ischemia, a feature that was confirmed by IHC results. Besides, the activation of A1ARs promoted the reduction of the brain lesion, as measured with T2W-MRI, and the improvement of neurological outcome including motor, sensory and reflex responses. These results show for the first time the in vivo PET imaging of A1ARs expression after cerebral ischemia in rats and the application of [18F]FLT to evaluate glial proliferation in response to treatment.
Conclusion: Notably, these data provide evidence for A1ARs playing a key role in the control of both the activation of resident glia and the de novo proliferation of microglia and macrophages after experimental stroke in rats.
Keywords: [18F]CPFPX, [18F]DPA-714, [18F]FLT, PET, A1ARs, cerebral ischemia, MRI, neuroinflammation
 Global reach, higher impact
Global reach, higher impact