Impact Factor
Theranostics 2021; 11(2):491-505. doi:10.7150/thno.51215 This issue Cite
Research Paper
Inter and intra-tumor somatostatin receptor 2 heterogeneity influences peptide receptor radionuclide therapy response
1. Department of Radiology and Nuclear Medicine, Erasmus MC Rotterdam, The Netherlands
2. Department of Molecular Genetics, Erasmus MC Rotterdam, The Netherlands
3. Oncode Institute, Erasmus MC Rotterdam, The Netherlands
4. Advanced Accelerator Applications SA, a Novartis Company, Erasmus MC Rotterdam, The Netherlands
Abstract
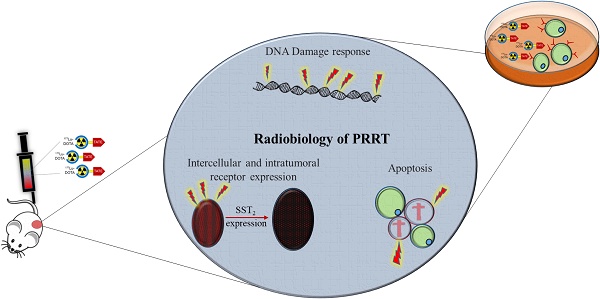
Patients with neuroendocrine tumors (NETs) can be treated with peptide receptor radionuclide therapy (PRRT). Here, the somatostatin analogue octreotate radiolabeled with lutetium-177 is targeted to NET cells by binding to the somatostatin receptor subtype 2 (SST2). During radioactive decay, DNA damage is induced, leading to NET cell death. Although the therapy proves to be effective, mortality rates remain high. To appropriately select more optimal treatment strategies, it is essential to first better understand the radiobiological responses of tumor cells to PRRT.
Methods: We analyzed PRRT induced radiobiological responses in SST2 expressing cells and xenografted mice using SPECT/MRI scanning and histological and molecular analyses. We measured [177Lu]Lu-DOTA-TATE uptake and performed analyses to visualize induction of DNA damage, cell death and other cellular characteristics.
Results: The highest accumulation of radioactivity was measured in the tumor and kidneys. PRRT induced DNA damage signaling and repair in a time-dependent manner. We observed intra-tumor heterogeneity of DNA damage and apoptosis, which was not attributed to proliferation or bioavailability. We found a strong correlation between high DNA damage levels and high SST2 expression. PRRT elicited a different therapeutic response between models with different SST2 expression levels. Heterogeneous SST2 expression levels were also confirmed in patient NETs.
Conclusion: Heterogeneous SST2 expression levels within NETs cause differentially induced DNA damage levels, influence recurrent tumor phenotypes and impact the therapeutic response in different models and potentially in patients. Our results contribute to a better understanding of PRRT effects, which might impact future therapeutic outcome of NET patients.
Keywords: Peptide receptor radionuclide therapy, neuroendocrine tumors, somatostatin receptor subtype 2, radiobiology, DNA damage response
 Global reach, higher impact
Global reach, higher impact