13.3
Impact Factor
Theranostics 2021; 11(2):522-539. doi:10.7150/thno.49426 This issue Cite
Research Paper
Melanocortin 1 receptor attenuates early brain injury following subarachnoid hemorrhage by controlling mitochondrial metabolism via AMPK/SIRT1/PGC-1α pathway in rats
1. Department of Neurosurgery, The Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310009, Zhejiang, China.
2. Department of Physiology & Pharmacology Loma Linda University, Loma Linda, CA 92350, USA.
3. Department of Neurosurgery Loma Linda University, Loma Linda, CA 92350, USA.
4. Department of Neurosurgery, Guangxi Medical University Cancer Hospital, 71 He Di Road, Nanning, 530021, Guangxi Zhuang Autonomous Region, China.
5. Department of Emergency Medicine, Bursa Yuksek Ihtisas Training and Research Hospital, University of Health Sciences, Bursa 16310, Turkey.
6. Department of Emergency Medicine, Bursa City Hospital, Bursa 16110, Turkey.
7. Burrell College of Osteopathic Medicine, New Mexico State University, Las Cruces, NM, USA.
Weilin Xu and Jun Yan have equally contributed to this work as co-first authors.
Abstract
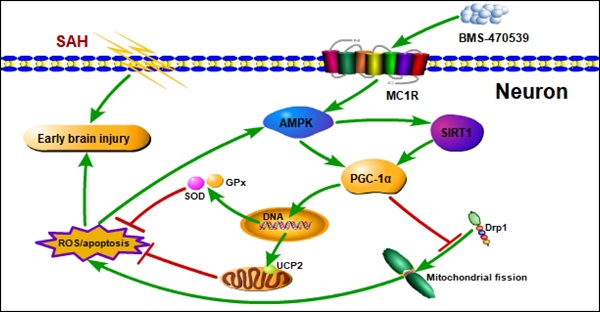
Mitochondria-mediated oxidative stress and apoptosis contribute greatly to early brain injury (EBI) following subarachnoid hemorrhage (SAH). This study hypothesized that activation of melanocortin 1 receptor (MC1R), using BMS-470539, attenuates EBI by controlling mitochondrial metabolism after SAH.
Methods: We utilized BMS-470539, MSG-606, selisistat, and PGC-1α to verify the neuroprotective effects of MC1R. We evaluated short- and long-term neurobehavior after SAH. Western blotting, immunofluorescence, and Golgi staining techniques were performed to assess changes in protein levels.
Results: The results of western blotting suggested that the expression of SIRT1 and PGC-1α were increased, reaching their peaks at 24 h following SAH. Moreover, BMS-470539 treatment notably attenuated neurological deficits, and also reduced long-term spatial learning and memory impairments caused by SAH. The underlying neuroprotective mechanisms of the BMS-470539/MC1R system were mediated through the suppression of oxidative stress, apoptosis, and mitochondrial fission by increasing the levels of SIRT1, PGC-1α, UCP2, SOD, GPx, Bcl-2, cyto-Drp1, and ATP, while decreasing the levels of cleaved caspase-3, Bax, mito-Drp1, ROS, GSH/GSSG, and NADPH/NADP+ ratios. The neuroprotective effects of the BMS-470539/MC1R system were significantly abolished by MSG-606, selisistat, and PGC-1α siRNA.
Conclusions: The activation of MC1R with BMS-470539 significantly attenuated EBI after SAH by suppressing the oxidative stress, apoptosis, and mitochondrial fission through the AMPK/SIRT1/PGC-1α signaling pathway.
Keywords: melanocortin 1 receptor, mitochondrial fission, oxidative stress, peroxisome proliferator-activated receptor gamma coactivator 1-alpha, subarachnoid hemorrhage
 Global reach, higher impact
Global reach, higher impact