13.3
Impact Factor
Theranostics 2021; 11(2):567-578. doi:10.7150/thno.48620 This issue Cite
Research Paper
CXCR4 PET imaging of mantle cell lymphoma using [68Ga]Pentixafor: comparison with [18F]FDG-PET
1. Dept. of Biomedical Imaging and Image-guided Therapy, Division of General and Pediatric Radiology, Medical University of Vienna, Austria
2. Department of Radiology, Memorial Sloan Kettering Cancer Center, New York, USA
3. Dept. of Internal Medicine I, Division of Oncology, Medical University of Vienna
4. Dept. of Biomedical Imaging and Image-guided Therapy, Division of Nuclear Medicine, Medical University of Vienna, Austria
5. Dept. of Internal Medicine I, Division of Hematology and Hemostaseology, Medical University of Vienna
6. Pharmaceutical Radiochemistry, Technical University of Munich, Garching, Germany
7. Clinical Institute of Pathology, Medical University of Vienna
Abstract
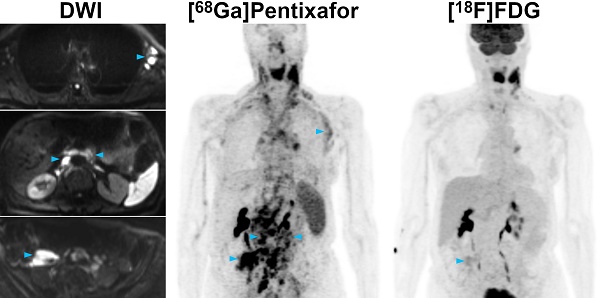
For PET imaging of mantle cell lymphoma (MCL), [18F]FDG (2-deoxy-2-[18F]fluoro-D-glucose) is the currently recommended radiotracer, although uptake is variable and bone marrow evaluation is limited. In this prospective study, we evaluated the novel CXCR4 (G-protein-coupled C-X-C chemokine receptor type 4) tracer [68Ga]Pentixafor in MCL patients, and compared it to [18F]FDG.
Methods: MCL patients underwent [68Ga]Pentixafor-PET/MRI, and, if required for routine purposes, also [18F]FDG-PET/MRI, before treatment. PET was evaluated separately for 23 anatomic regions (12 lymph node stations and 11 organs/tissues), using MRI as the main reference standard. Standardized uptake values (SUVmax and SUVmean) and tumor-to-background ratios (TBRblood and TBRliver) were calculated. General Estimation Equations (GEE) were used to compare [68Ga]Pentixafor-PET and [18F]FDG-PET sensitivities and positive predictive values (PPV). For bone marrow involvement, where biopsy served as the main reference standard, and splenic involvement, receiver operating characteristic curves were used to determine the optimal SUV and TBR cut-off values, and areas under the curve (AUC) were calculated.
Results: Twenty-two MCL patients were included. [68Ga]Pentixafor-PET sensitivity (100%) was significantly higher than for [18F]FDG-PET (75.2%) (P<0.001), and PPV was slightly, but not significantly lower (94.0%.vs. 96.5%; P=0.21). SUVs and TBRs were significantly higher for [68Ga]Pentixafor-PET than for [18F]FDG-PET (P<0.001 in all cases); the greatest difference was observed for mean TBRblood, with 4.9 for [68Ga]Pentixafor-PET and 2.0 for [18F]FDG-PET. For bone marrow involvement, [68Ga]Pentixafor-PET SUVmean showed an AUC of 0.92; and for splenic involvement, TBRblood showed an AUC of 0.81.
Conclusion: [68Ga]Pentixafor-PET may become an alternative to [18F]FDG-PET in MCL patients, showing clearly higher detection rates and better tumor-to-background contrast.
Keywords: Lymphoma, Chemokine receptor, CXCR4, Positron emission tomography, Magnetic resonance imaging
 Global reach, higher impact
Global reach, higher impact