13.3
Impact Factor
Theranostics 2021; 11(2):700-714. doi:10.7150/thno.47800 This issue Cite
Research Paper
PLAGL2 promotes the proliferation and migration of gastric cancer cells via USP37-mediated deubiquitination of Snail1
1. Department of Gastrointestinal Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Jiefang Road No. 1277, Wuhan 430022, China.
2. Department of Emergency Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Jiefang Road No. 1277, Wuhan 430022, China.
3. Department of Gastrointestinal Surgery II, Renmin Hospital of Wuhan University, Zhang Zhidong Road No. 99 Wuhan 430060, China.
*These authors contributed equally to this work.
Abstract
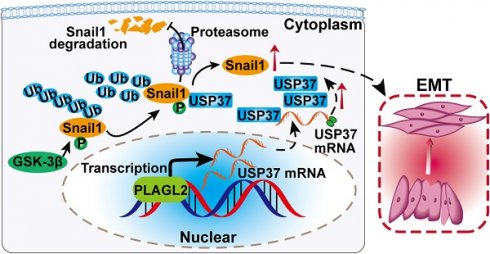
Rationale: PLAGL2 (pleomorphic adenoma gene like-2), a zinc finger PLAG transcription factor, is aberrantly expressed in several malignant tumors. However, the biological roles of PLAGL2 and its underlying mechanism in gastric cancer (GC) remain unclear.
Methods: A series of experiments in vitro and in vivo were conducted to reveal the role of PLAGL2 in GC progression.
Results: The data revealed that PLAGL2 promotes GC cell proliferation, migration, invasion, and EMT in vitro and in vivo. Mechanistically, we demonstrated the critical role of PLAGL2 in the stabilization of snail family transcriptional repressor 1 (Snail1) and promoting Snail1-mediated proliferation and migration of GC cells. PLAGL2 activated the transcription of deubiquitinase USP37, which then interacted with and deubiquitinated Snail1 protein directly. In addition, GSK-3β-dependent phosphorylation of Snail1 protein is essential for USP37-mediated Snail1 deubiquitination regulation.
Conclusions: In general, PLAGL2 promotes the proliferation and migration of GC cells through USP37-mediated deubiquitination of Snail1 protein. This work provided potential therapeutic targets for GC treatment.
Keywords: PLAGL2, USP37, Snail1, GC, deubiquitination
 Global reach, higher impact
Global reach, higher impact