Impact Factor
Theranostics 2021; 11(3):1295-1309. doi:10.7150/thno.50150 This issue Cite
Research Paper
Blocking NHE1 stimulates glioma tumor immunity by restoring OXPHOS function of myeloid cells
1. Department of Neurology and Pittsburgh Institute for Neurodegenerative Disease, University of Pittsburgh, Pittsburgh, PA 15213.
2. Animal Imaging Center, University of Pittsburgh, Pittsburgh, Pennsylvania, 15213.
3. Department of Neurological Surgery, University of Pittsburgh, Pittsburgh, PA 15213.
4. Department of Neurobiology, University of Pittsburgh, Pittsburgh, Pennsylvania, 15213.
5. Department of Neurosurgery, University of Michigan Medical School, Ann Arbor, MI, USA.
6. Department of Neurology, First Affiliated Hospital, Harbin Medical University, Harbin, Heilongjiang, China.
7. Veterans Affairs Pittsburgh Health Care System, Geriatric Research, Educational and Clinical Center, Pittsburgh, Pennsylvania.
Abstract
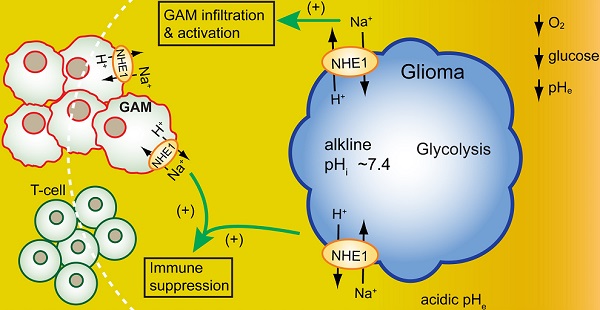
Background: Immunosuppressive tumor microenvironment (TME) in glioblastoma (GBM) is one of the contributing factors for failed immunotherapies. Therefore, there is an urgent need to better understand TME and to identify novel modulators of TME for more effective GBM therapies. We hypothesized that H+ extrusion protein Na/H exchanger 1 (NHE1) plays a role in dysregulation of glucose metabolism and immunosuppression of GBM. We investigated the efficacy of blockade of NHE1 activity in combination with temozolomide (TMZ) therapy in increasing anti-tumor immunity.
Methods: Mouse syngeneic intracranial glioma model was used to test four treatment regimens: DMSO (Vehicle-control), TMZ, NHE1 specific inhibitor HOE642, or TMZ+HOE642 (T+H) combination. Ex vivo 1H/19Fluorine magnetic resonance imaging (MRI) with cell tracking agent Vsense was performed to monitor the infiltration of glioma-associated microglia/myeloid cells (GAMs). Glucose metabolism and transcriptome profiles were analyzed by Seahorse analyzer and bulk RNA-sequencing. The impact of selective Nhe1 deletion in GAMs on sensitivity to anti-PD-1 therapy was evaluated in transgenic NHE1 knockout (KO) mice.
Results: Among the tested treatment regimens, the T+H combination therapy significantly stimulated the infiltration of GAMs and T-cells; up-regulated Th1 activation, and mitochondrial oxidative phosphorylation (OXPHOS) pathway genes, increased glucose uptake and mitochondrial mass, and decreased aerobic glycolysis in GAMs. Selective deletion of Nhe1 in Cx3cr1+ Nhe1 KO mice increased anti-tumor immunity and sensitivity to TMZ plus anti-PD-1 combinatorial therapy.
Conclusions: NHE1 plays a role in developing glioma immunosuppressive TME in part by dysregulating glucose metabolism of GAMs and emerges as a therapeutic target for improving glioma immunity.
Keywords: anti-PD-1 therapy, glycolysis, immunosuppression, temozolomide, tumor-associated microglia/macrophage
 Global reach, higher impact
Global reach, higher impact