Impact Factor
Theranostics 2021; 11(3):1429-1445. doi:10.7150/thno.45351 This issue Cite
Research Paper
Breast cancer exosomes contribute to pre-metastatic niche formation and promote bone metastasis of tumor cells
1. State Key Laboratory of Agrobiotechnology, College of Biological Sciences, China Agricultural University, Beijing 100193, China.
2. State Key Lab of Space Medicine Fundamentals and Application, China Astronaut Research and Training Center, Beijing 100094, China.
3. Department of Oncology, Chinese PLA General Hospital, Beijing 100853, China.
#Equal contributions to this work.
Abstract
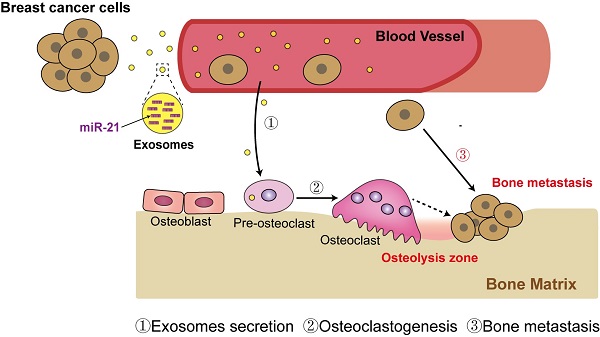
Rationale: Breast cancer preferentially develops osteolytic bone metastasis, which makes patients suffer from pain, fractures and spinal cord compression. Accumulating evidences have shown that exosomes play an irreplaceable role in pre-metastatic niche formation as a communication messenger. However, the function of exosomes secreted by breast cancer cells remains incompletely understood in bone metastasis of breast cancer.
Methods: Mouse xenograft models and intravenous injection of exosomes were applied for analyzing the role of breast cancer cell-derived exosomes in vivo. Effects of exosomes secreted by the mildly metastatic MDA231 and its subline SCP28 with highly metastatic ability on osteoclasts formation were confirmed by TRAP staining, ELISA, microcomputed tomography, histomorphometric analyses, and pit formation assay. The candidate exosomal miRNAs for promoting osteoclastogenesis were globally screened by RNA-seq. qRT-PCR, western blot, confocal microscopy, and RNA interfering were performed to validate the function of exosomal miRNA.
Results: Implantation of SCP28 tumor cells in situ leads to increased osteoclast activity and reduced bone density, which contributes to the formation of pre-metastatic niche for tumor cells. We found SCP28 cells-secreted exosomes are critical factors in promoting osteoclast differentiation and activation, which consequently accelerates bone lesion to reconstruct microenvironment for bone metastasis. Mechanistically, exosomal miR-21 derived from SCP28 cells facilitates osteoclastogenesis through regulating PDCD4 protein levels. Moreover, miR-21 level in serum exosomes of breast cancer patients with bone metastasis is significantly higher than that in other subpopulations.
Conclusion: Our results indicate that breast cancer cell-derived exosomes play an important role in promoting breast cancer bone metastasis, which is associated with the formation of pre-metastatic niche via transferring miR-21 to osteoclasts. The data from patient samples further reflect the significance of miR-21 as a potential target for clinical diagnosis and treatment of breast cancer bone metastasis.
Keywords: exosomes, pre-metastatic niche, osteoclasts, miR-21, bone metastasis
 Global reach, higher impact
Global reach, higher impact