Impact Factor
Theranostics 2021; 11(4):1626-1640. doi:10.7150/thno.51507 This issue Cite
Research Paper
Metastatic colorectal cancer cells maintain the TGFβ program and use TGFBI to fuel angiogenesis
1. Cancer Research Institute of Montpellier, Tumor Microenvironment and Resistance to Treatment Laboratory, INSERM U1194, Montpellier, France.
2. Institut National de la Santé et de la Recherche Médicale, Montpellier, France.
3. Institut du Cancer de Montpellier, Montpellier, France.
4. Université de Montpellier, Montpellier, France.
5. Metastasis Research Laboratory, GIGA Cancer, University of Liège, Liège, Belgium.
6. University of Brescia, Department of Molecular and Translational Medicine, Brescia, Italy.
7. Cancer Research Institute of Montpellier, Cancer Bioinformatics and Systems Biology Team, INSERM U1194, Montpellier, France.
8. Institut de Génomique Fonctionnelle, Montpellier, France.
9. Centre National de la Recherche Scientifique, Montpellier, France.
10. Center for Microscopy and Molecular Imaging (CMMI), Université libre de Bruxelles (ULB), rue Adrienne Bolland 8, B-6041 Charleroi (Gosselies), Belgium.
11. Nuclear Medicine department, ULB Hôpital Érasme, route de Lennik 808, B-1070 Brussels, Belgium.
12. Gunma University Initiative for Advanced Research, International Open Laboratory, Universities of Liege and Montpellier Laboratory, Gunma University, Gunma, Japan.
13. Cancer Research Institute of Montpellier, Hormone Signaling and Cancer Laboratory, Montpellier, France.
14. Cancer Research Institute of Montpellier, Oncogenic Pathways in Cancer Laboratory, INSERM U1194, Montpellier, France.
15. GIGA-Viral Vectors Platform, University of Liège, Liège, Belgium.
16. BIOTHEQUE, University of Liege, Liege, Belgium.
17. Department of Abdominal Surgery, University Hospital, University of Liège, Liège, Belgium.
18. Department of Medical Oncology, University Hospital, University of Liège, Liège, Belgium.
19. Department of Pathology, University Hospital, University of Liège, Liège, Belgium.
*Equal contributions to this work.
Abstract
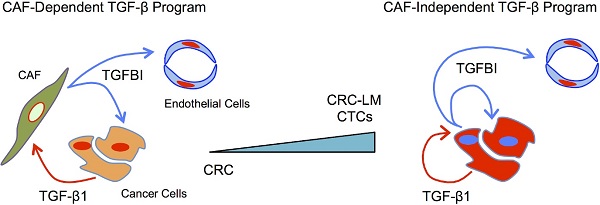
Colorectal cancer (CRC) cells are traditionally considered unresponsive to TGFβ due to mutations in the receptors and/or downstream signaling molecules. TGFβ influences CRC cells only indirectly via stromal cells, such as cancer-associated fibroblasts. However, CRC cell ability to directly respond to TGFβ currently remains unexplored. This represents a missed opportunity for diagnostic and therapeutic interventions.
Methods: We examined whether cancer cells from primary CRC and liver metastases respond to TGFβ by inducing TGFβ-induced protein ig-h3 (TGFBI) expression, and the contribution of canonical and non-canonical TGFβ signaling pathways to this effect. We then investigated in vitro and in vivo TGFBI impact on metastasis formation and angiogenesis. Using patient serum samples and an orthotopic mouse model of CRC liver metastases we assessed the diagnostic/tumor targeting value of novel antibodies against TGFBI.
Results: Metastatic CRC cells, such as circulating tumor cells, directly respond to TGFβ. These cells were characterized by the absence of TGFβ receptor mutations and the frequent presence of p53 mutations. The pro-tumorigenic program orchestrated by TGFβ in CRC cells was mediated through TGFBI, the expression of which was positively regulated by non-canonical TGFβ signaling cascades. TGFBI inhibition was sufficient to significantly reduce liver metastasis formation in vivo. Moreover, TGFBI pro-tumorigenic function was linked to its ability to stimulate angiogenesis. TGFBI levels were higher in serum samples from untreated patients with CRC than in patients who were receiving chemotherapy. A radiolabeled anti-TGFBI antibody selectively targeted metastatic lesions in vivo, underscoring its diagnostic and therapeutic potential.
Conclusions: TGFβ signaling in CRC cells directly contributes to their metastatic potential and stromal cell-independence. Proteins downstream of activated TGFβ, such as TGFBI, represent novel diagnostic and therapeutic targets for more specific anti-metastatic therapies.
Keywords: alternative TGFβ signaling, liver metastases, endothelial cells
 Global reach, higher impact
Global reach, higher impact