13.3
Impact Factor
Theranostics 2021; 11(5):2020-2033. doi:10.7150/thno.47269 This issue Cite
Research Paper
Imaging temozolomide-induced changes in the myeloid glioma microenvironment
1. European Institute for Molecular Imaging - EIMI, University of Münster, Münster, Germany.
2. PET Imaging in Drug Design and Development (PET3D), Münster, Germany.
3. Tecnomed Foundation and Medicine and Surgery Department, University of Milano-Bicocca, Milan, Italy.
4. Experimental Imaging Center, IRCCS San Raffaele Scientific Institute, Milan, Italy.
5. Institute of Molecular Bioimaging and Physiology (IBFM), CNR, Segrate (MI), Italy.
6. Université Paris-Saclay, CEA, CNRS, Inserm, BioMaps, Orsay, France.
7. Department of Nuclear Medicine, University Hospital Münster, Münster, Germany.
8. Department of Neurosurgery, University Hospital Münster, Münster, Germany.
9. Campus Chemical Instrument Center, Mass Spectrometry and Proteomics Facility (CCIC-MSP), The Ohio State University, Columbus, OH, United States.
10. Cells in Motion (CiM) Cluster of Excellence, Münster, Germany.
11. Department of Neurology, University Hospital Münster, Münster, Germany.
12. Imaging Neuroinflammation in Neurodegenerative Diseases (INMIND) EU FP7 consortium, Münster, Germany.
13. Department of Geriatrics, Johanniter Hospital, Evangelische Kliniken, Bonn, Germany.
#These authors contributed equally to this work.
Abstract
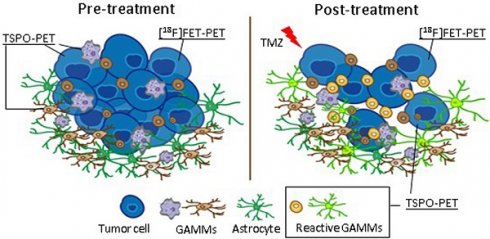
Rationale: The heterogeneous nature of gliomas makes the development and application of novel treatments challenging. In particular, infiltrating myeloid cells play a role in tumor progression and therapy resistance. Hence, a detailed understanding of the dynamic interplay of tumor cells and immune cells in vivo is necessary. To investigate the complex interaction between tumor progression and therapy-induced changes in the myeloid immune component of the tumor microenvironment, we used a combination of [18F]FET (amino acid metabolism) and [18F]DPA-714 (TSPO, GAMMs, tumor cells, astrocytes, endothelial cells) PET/MRI together with immune-phenotyping. The aim of the study was to monitor temozolomide (TMZ) treatment response and therapy-induced changes in the inflammatory tumor microenvironment (TME).
Methods: Eighteen NMRInu/nu mice orthotopically implanted with Gli36dEGFR cells underwent MRI and PET/CT scans before and after treatment with TMZ or DMSO (vehicle). Tumor-to-background (striatum) uptake ratios were calculated and areas of unique tracer uptake (FET vs. DPA) were determined using an atlas-based volumetric approach.
Results: TMZ therapy significantly modified the spatial distribution and uptake of both tracers. [18F]FET uptake was significantly reduced after therapy (-53 ± 84%) accompanied by a significant decrease of tumor volume (-17 ± 6%). In contrast, a significant increase (61 ± 33%) of [18F]DPA-714 uptake was detected by TSPO imaging in specific areas of the tumor. Immunohistochemistry (IHC) validated the reduction in tumor volumes and further revealed the presence of reactive TSPO-expressing glioma-associated microglia/macrophages (GAMMs) in the TME.
Conclusion: We confirm the efficiency of [18F]FET-PET for monitoring TMZ-treatment response and demonstrate that in vivo TSPO-PET performed with [18F]DPA-714 can be used to identify specific reactive areas of myeloid cell infiltration in the TME.
Keywords: glioblastoma, temozolomide, GAMM, [18F]DPA-714, [18F]FET, TSPO
 Global reach, higher impact
Global reach, higher impact