Impact Factor
Theranostics 2021; 11(6):2564-2580. doi:10.7150/thno.45280 This issue Cite
Research Paper
IL20RA signaling enhances stemness and promotes the formation of an immunosuppressive microenvironment in breast cancer
1. School of Medicine, Nankai University, 94 Weijin Road, Tianjin, PR China.
2. Tianjin's Clinical Research Center for Cancer, Tianjin, PR China.
3. Key Laboratory of Breast Cancer Prevention and Therapy, Tianjin Medical University, Ministry of Education, Tianjin, PR China.
4. Department of Blood Transfusion, Tianjin Medical University Cancer Institute and Hospital, Tianjin, PR China.
5. Department of Colorectal Surgery, Tianjin Union Medical Centre, Tianjin, PR China.
6. Tianjin Key Laboratory of Tumour Microenvironment and Neurovascular Regulation, Tianjin, PR China.
Abstract
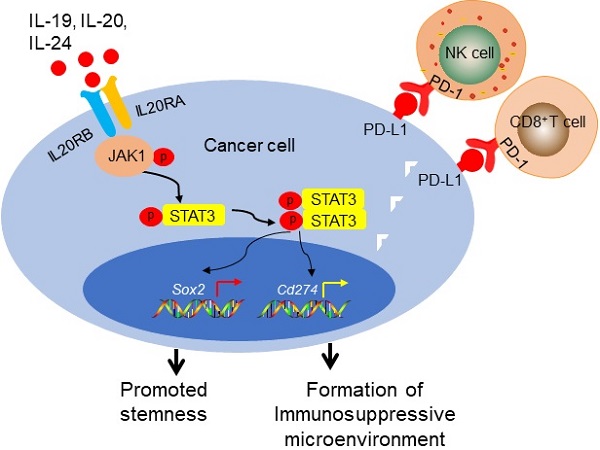
Rationale: Tumor microenvironment interacts with tumor cells to regulate their stemness properties through various cytokines and cytokine receptors. Previous studies revealed the possible role of interleukin 20 receptor subunit alpha (IL20RA) signaling in the progression of several types of tumors. However, its regulatory effects on the stemness and the microenvironment of breast cancer need to be studied.
Methods: Immunohistochemical staining and western blot analysis were used to evaluate the association between IL20RA and SOX2 in breast tumors and noncancerous tissues. Enzyme-linked immunosorbent assay and TCGA dataset analysis were performed to determine the function of IL20RA signaling in breast cancer progression. Gain- and loss-of-function methods were performed to examine the effects of IL20RA on the stemness of breast cancer cells. The stemness features were analyzed by detecting the expression of core stemness genes, side population (SP), sphere formation ability, and aldehyde dehydrogenase (ALDH) activity. Flow cytometric analysis was applied to detect the changes of tumor-infiltration lymphocytes in tumor tissues in mice. Based on the relevant molecular mechanisms elucidated in this study, a novel IL20RA-targeted liposomal nanoparticle encapsulating the signal transducer and activator of transcription 3 (STAT3) inhibitor stattic (NP-Stattic-IL20RA) was synthesized. These NPs were combined with anti-programmed death ligand 1 (PD-L1) antibody and chemotherapy to inhibit the development of breast tumors in mice.
Results: IL20RA is highly expressed in human breast cancers and is positively associated with the SOX2 expression. IL20RA increases the SP and ALDHbr proportions of breast cancer cells, enhances the sphere formation ability, and promotes the expression of core stemness genes, such as Sox2 and Oct4, as well as increases chemoresistance of breast cancer cells. IL20RA promotes the tumor-initiating ability and lung metastasis of breast cancer cells in vivo. In addition, IL20RA activates the Janus kinase 1 (JAK1)-STAT3-SOX2 signaling pathway, leading to increased expression of PD-L1 and reduced recruitment of anti-cancer lymphocytes, including CD8+ T cells and natural killer cells. Meanwhile, IL20RA signaling enhances the proportion of myeloid-derived suppressor cells. Combined with anti-PD-L1 antibody and NPs-Stattic-IL20RA, the chemotherapeutic efficacy was increased in breast cancer mouse models in vivo.
Conclusion: Collectively, our results reveal that the IL20RA pathway is a novel signaling pathway involved in promoting the stemness features of breast cancer along with the formation of a tumor-favorable immune microenvironment. Targeting the IL20RAhi population with STAT3 signaling inhibition combined with anti-PD-L1 antibody can increase the therapeutic efficacy of chemotherapeutic agents for breast cancer. This study thus introduces a promising novel strategy for breast cancer therapy.
Keywords: IL20RA, SOX2, breast cancer, stemness, nanoparticles
 Global reach, higher impact
Global reach, higher impact