13.3
Impact Factor
Theranostics 2021; 11(6):2788-2805. doi:10.7150/thno.53330 This issue Cite
Research Paper
Lipin 1 deficiency causes adult-onset myasthenia with motor neuron dysfunction in humans and neuromuscular junction defects in zebrafish
1. Key laboratory of Resource Biology and Biotechnology in Western China, Ministry of Education, School of Medicine, Northwest University, Xi'an 710069, China.
2. Institute of Oceanography, Minjiang University, Fuzhou 350108, China.
3. State Key Laboratory of Photon-Technology in Western China Energy, Institute of Photonics and Photon-Technology, Northwest University, Xi'an 710069, China.
4. Institute of Biochemical Sciences, College of Life Science, National Taiwan University, Taipei, Taiwan.
5. Department of Life Science, Tunghai University, Taichung, Taiwan.
6. Sino-French Hoffmann Institute, School of Basic Medical Sciences, Guangzhou Medical University, Guangzhou 511436, China.
#These authors contributed equally to this work.
Abstract
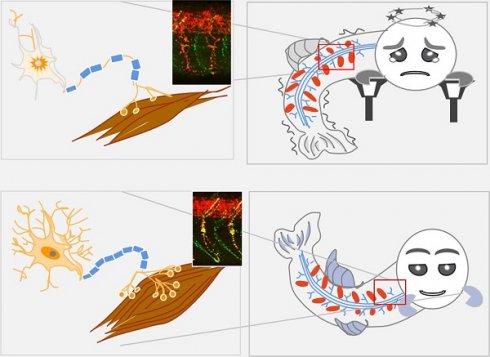
Lipin 1 is an intracellular protein acting as a phosphatidic acid phosphohydrolase enzyme controlling lipid metabolism. Human recessive mutations in LPIN1 cause recurrent, early-onset myoglobinuria, a condition normally associated with muscle pain and weakness. Whether and how lipin 1 deficiency in humans leads to peripheral neuropathy is yet unclear. Herein, two novel compound heterozygous mutations in LPIN1 with neurological disorders, but no myoglobinuria were identified in an adult-onset syndromic myasthenia family. The present study sought to explore the pathogenic mechanism of LPIN1 in muscular and neural development.
Methods: The clinical diagnosis of the proband was compared to the known 48 cases of LPIN1 recessive homozygous mutations. Whole-exome sequencing was carried out on the syndromic myasthenia family to identify the causative gene. The pathogenesis of lipin 1 deficiency during somitogenesis and neurogenesis was investigated using the zebrafish model. Whole-mount in situ hybridization, immunohistochemistry, birefringence analysis, touch-evoke escape response and locomotion assays were performed to observe in vivo the changes in muscles and neurons. The conservatism of the molecular pathways regulated by lipin 1 was evaluated in human primary glioblastoma and mouse myoblast cells by siRNA knockdown, drug treatment, qRT-PCR and Western blotting analysis.
Results: The patient exhibited adult-onset myasthenia accompanied by muscle fiber atrophy and nerve demyelination without myoglobinuria. Two novel heterozygous mutations, c.2047A>C (p.I683L) and c.2201G>A (p.R734Q) in LPIN1, were identified in the family and predicted to alter the tertiary structure of LPIN1 protein. Lipin 1 deficiency in zebrafish embryos generated by lpin1 morpholino knockdown or human LPIN1 mutant mRNA injections reproduced the myotomes defects, a reduction both in primary motor neurons and secondary motor neurons projections, morphological changes of post-synaptic clusters of acetylcholine receptors, and myelination defects, which led to reduced touch-evoked response and abnormalities of swimming behaviors. Loss of lipin 1 function in zebrafish and mammalian cells also exhibited altered expression levels of muscle and neuron markers, as well as abnormally enhanced Notch signaling, which was partially rescued by the specific Notch pathway inhibitor DAPT.
Conclusions: These findings pointed out that the compound heterozygous mutations in human LPIN1 caused adult-onset syndromic myasthenia with peripheral neuropathy. Moreover, zebrafish could be used to model the neuromuscular phenotypes due to the lipin 1 deficiency, where a novel pathological role of over-activated Notch signaling was discovered and further confirmed in mammalian cell lines.
Keywords: LPIN1, syndromic myasthenia, zebrafish morphants, neuromuscular development, Notch signaling
 Global reach, higher impact
Global reach, higher impact