13.3
Impact Factor
Theranostics 2021; 11(6):2845-2859. doi:10.7150/thno.51857 This issue Cite
Research Paper
Smad3 deficiency promotes beta cell proliferation and function in db/db mice via restoring Pax6 expression
1. Department of Medicine and Therapeutics, Li Ka Shing Institute of Health Sciences, The Chinese University of Hong Kong
2. Research Center for Integrated Chinese and Western Medicine, and Department of Cardiology, The Second Affiliated Hospital, Southwest Medical University, Luzhou, Sichuan, China
3. Department of Anatomical and Cellular Pathology, State Key Laboratory of Translational Oncology, The Chinese University of Hong Kong
4. State Key Laboratory of Bioelectronics, Jiangsu Key Laboratory for Biomaterials and Devices, School of Biological Sciences & Medical Engineering, Southeast University, Nanjing, China
5. School of life science, the Chinese University of Hong Kong, Hong Kong SAR, China
6. Guangdong-Hong Kong Joint Laboratory on Immunological and Genetic Kidney Disease, Guangdong Academy of Medical Sciences, Guangdong Provincial People's Hospital, Guangzhou, China.
* These authors contribute equality to the work.
Abstract
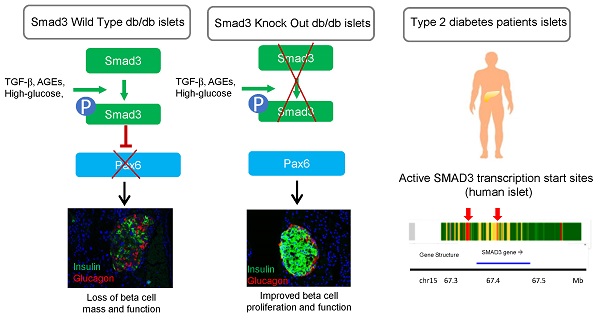
Rationale: Transforming Growth Factor-beta (TGF-β) /Smad3 signaling has been shown to play important roles in fibrotic and inflammatory diseases, but its role in beta cell function and type 2 diabetes is unknown.
Methods: The role of Smad3 in beta cell function under type 2 diabetes condition was investigated by genetically deleting Smad3 from db/db mice. Phenotypic changes of pancreatic islets and beta cell function were compared between Smad3 knockout db/db (Smad3KO-db/db) mice and Smad3 wild-type db/db (Smad3WT-db/db) mice, and other littermate controls. Islet-specific RNA-sequencing was performed to identify Smad3-dependent differentially expressed genes associated with type 2 diabetes. In vitro beta cell proliferation assay and insulin secretion assay were carried out to validate the mechanism by which Smad3 regulates beta cell proliferation and function.
Results: The results showed that Smad3 deficiency completely protected against diabetes-associated beta cell loss and dysfunction in db/db mice. By islet-specific RNA-sequencing, we identified 8160 Smad3-dependent differentially expressed genes associated with type 2 diabetes, where Smad3 deficiency markedly prevented the down-regulation of those genes. Mechanistically, Smad3 deficiency preserved the expression of beta cell development mediator Pax6 in islet, thereby enhancing beta cell proliferation and function in db/db mice in vivo and in Min6 cells in vitro.
Conclusions: Taken together, we discovered a pathogenic role of Smad3 in beta cell loss and dysfunction via targeting the protective Pax6. Thus, Smad3 may represent as a novel therapeutic target for type 2 diabetes prevention and treatment.
Keywords: Type 2 diabetes, Islet beta cells, Smad3, Pax6
 Global reach, higher impact
Global reach, higher impact