13.3
Impact Factor
Theranostics 2021; 11(6):2860-2875. doi:10.7150/thno.51797 This issue Cite
Research Paper
Cisplatin-resistant NSCLC cells induced by hypoxia transmit resistance to sensitive cells through exosomal PKM2
1. Department of Nuclear Medicine, Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200127, China
2. Shanghai Key Laboratory of Molecular Imaging, Shanghai University of Medicine and Health Sciences, Shanghai 201318, China
3. Department of Dermatology, Shanghai General Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200080, China
4. Department of Nuclear Medicine, Shanghai Chest Hospital, Shanghai Jiao Tong University, Shanghai 200030, China
Abstract
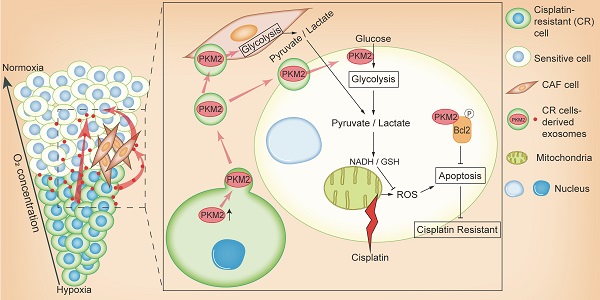
Hypoxia is commonly observed in solid tumors and contributes to the resistance of DNA damage drugs. However, the mechanisms behind this resistance are still unclear. In this study, we aimed to explore the effects of hypoxia-induced exosomes on non-small cell lung cancer (NSCLC).
Methods: NSCLC cells were subjected to either normoxic or hypoxic conditions to assess cell survival and changes in the expression levels of key proteins. Comparative proteomics were performed to identify exosomal PKM2 in normoxic or hypoxic cisplatin-resistant NSCLC cells-derived exosomes. Functions of hypoxia induced-exosomal PKM2 in promoting cisplatin resistance to NSCLC cells were evaluated both in vitro and in vivo experiments and the molecular mechanisms of hypoxia induced-exosomal PKM2 were demonstrated using flow cytometry, immunoblotting, oxidative stress detection and histological examination. A series of in vitro experiments were performed to evaluate the function of hypoxia-induced exosomes on cancer-associated fibroblasts (CAFs).
Results: Hypoxia exacerbated the cisplatin resistance in lung cancer cells due to the increased expression of PKM2 that was observed in the exosomes secreted by hypoxic cisplatin-resistance cells. We identified that hypoxia-induced exosomal PKM2 transmitted cisplatin-resistance to sensitive NSCLC cells in vitro and in vivo. Mechanistically, hypoxia-induced exosomal PKM2 promoted glycolysis in NSCLC cells to produce reductive metabolites, which may neutralize reactive oxygen species (ROS) induced by cisplatin. Additionally, hypoxia-induced exosomal PKM2 inhibited apoptosis in a PKM2-BCL2-dependent manner. Moreover, hypoxia-induced exosomal PKM2 reprogrammed CAFs to create an acidic microenvironment promoting NSCLC cells proliferation and cisplatin resistance.
Conclusions: Our findings revealed that hypoxia-induced exosomes transmit cisplatin resistance to sensitive NSCLC cells by delivering PKM2. Exosomal PKM2 may serve as a promising biomarker and therapeutic target for cisplatin resistance in NSCLC.
Keywords: Exosomes, NSCLC, Drug-resistance, PKM2, CAFs
 Global reach, higher impact
Global reach, higher impact