13.3
Impact Factor
Theranostics 2021; 11(7):3167-3182. doi:10.7150/thno.52028 This issue Cite
Research Paper
Holo-lactoferrin: the link between ferroptosis and radiotherapy in triple-negative breast cancer
1. Department of Nutrition and Food Hygiene, School of Public Health, Soochow University, 199 Renai Road, Suzhou 215123, China
2. Center of Child Health Management, Children's Hospital of Soochow University, Suzhou, 215025, China
3. Laboratory Center, Medical College of Soochow University, Suzhou, 215123, China
4. State Key Laboratory of Radiation Medicine and Protection, Collaborative Innovation Center of Radiation Medicine of Jiangsu Higher Education Institutions, School of Radiation Medicine and Protection, Soochow University, 199 Renai Road, Suzhou 215123, China
Abstract
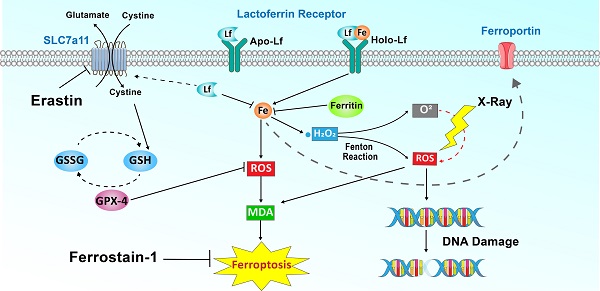
Rationale: Iron-saturated Lf (Holo-Lactoferrin, Holo-Lf) exhibits a superior anticancer property than low iron-saturated Lf (Apo-Lf). Ferroptosis is an iron-dependent cell death characterized by the accumulation of lipid peroxidation products and lethal reactive oxygen species (ROS). Radiotherapy also exerts its therapeutic effect through ROS.
Methods: The effect of different iron-saturated Lf on ferroptosis and radiotherapy were tested on triple-negative breast cancer (TNBC) cell line MDA-MB-231 and non-TNBC cell line MCF-7.
Results: Holo-Lf significantly increased the total iron content, promoted ROS generation, increased lipid peroxidation end product, malondialdehyde (MDA), and enhanced ferroptosis of MDA-MB-231 cells. By contrast, Apo-Lf upregulated SLC7a11 expression, increased GSH generation and inhibited ferroptosis of MDA-MB-231 cells. However, non-TNBC MCF-7 cells were resistant to Holo-Lf-induced ferroptosis because MCF-7 cells have a higher redox balance capacity than MDA-MB-231 cells. More importantly, Holo-Lf downregulated HIF-1α expression, ameliorated the hypoxia microenvironment in subcutaneous MDA-MB-231 tumors, and promoted radiation-induced DNA damage to hypoxic MDA-MB-231 cells. Finally, the efficacy of radiotherapy to MDA-MB-231 tumors was enhanced by Holo-Lf.
Conclusion: Holo-Lf could induce ferroptosis in MDA-MB-231 cells and sensitize MDA-MB-231 tumors to radiotherapy.
Keywords: Holo-Lf, ferroptosis, radiotherapy, triple-negative breast cancer, hypoxia
 Global reach, higher impact
Global reach, higher impact