Impact Factor
Theranostics 2021; 11(7):3229-3243. doi:10.7150/thno.55921 This issue Cite
Research Paper
Large-scale tumor-associated collagen signatures identify high-risk breast cancer patients
1. Key Laboratory of OptoElectronic Science and Technology for Medicine of Ministry of Education, Fujian Provincial Key Laboratory of Photonics Technology, Fujian Normal University, Fuzhou, China
2. Breast Surgery Ward, Department of General Surgery, Fujian Medical University Union Hospital, Fuzhou, China
3. Department of Pathology, Fujian Medical University Union Hospital, Fuzhou, China
4. Department of Radiation Oncology, Harbin Medical University Cancer Hospital, Harbin, China
5. College of Physics and Electronic Information Engineering, Minjiang University, Fuzhou, China
6. Department of Ophthalmology and Optometry, Fujian Medical University, Fuzhou, China
7. College of Life Science, Fujian Normal University, Fuzhou, China
8. Department of Medical Oncology, Harbin Medical University Cancer Hospital, Harbin, China
9. National Center for Supercomputing Applications, University of Illinois at Urbana-Champaign, Urbana, USA
10. Beckman Institute for Advanced Science and Technology, University of Illinois at Urbana-Champaign, Urbana, USA
†These authors contributed equally to this work.
Abstract
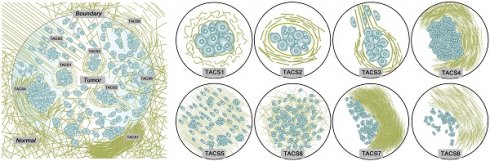
The notion of personalized medicine demands proper prognostic biomarkers to guide the optimal therapy for an invasive breast cancer patient. However, various risk prediction models based on conventional clinicopathological factors and emergent molecular assays have been frequently limited by either a low strength of prognosis or restricted applicability to specific types of patients. Therefore, there is a critical need to develop a strong and general prognosticator.
Methods: We observed five large-scale tumor-associated collagen signatures (TACS4-8) obtained by multiphoton microscopy at the invasion front of the breast primary tumor, which contrasted with the three tumor-associated collagen signatures (TACS1-3) discovered by Keely and coworkers at a smaller scale. Highly concordant TACS1-8 classifications were obtained by three independent observers. Using the ridge regression analysis, we obtained a TACS-score for each patient based on the combined TACS1-8 and established a risk prediction model based on the TACS-score. In a blind fashion, consistent retrospective prognosis was obtained from 995 breast cancer patients in both a training cohort (n= 431) and an internal validation cohort (n = 300) collected from one clinical center, and in an external validation cohort (n = 264) collected from a different clinical center.
Results: TACS1-8 model alone competed favorably with all reported models in predicting disease-free survival (AUC: 0.838, [0.800-0.872]; 0.827, [0.779-0.868]; 0.807, [0.754-0.853] in the three cohorts) and stratifying low- and high-risk patients (HR 7.032, [4.869-10.158]; 6.846, [4.370-10.726], 4.423, [2.917-6.708]). The combination of these factors with the TACS-score into a nomogram model further improved the prognosis (AUC: 0.865, [0.829-0.896]; 0.861, [0.816-0.898]; 0.854, [0.805-0.894]; HR 7.882, [5.487-11.323]; 9.176, [5.683-14.816], and 5.548, [3.705-8.307]). The nomogram identified 72 of 357 (~20%) patients with unsuccessful 5-year disease-free survival that might have been undertreated postoperatively.
Conclusions: The risk prediction model based on TACS1-8 considerably outperforms the contextual clinical model and may thus convince pathologists to pursue a TACS-based breast cancer prognosis. Our methodology identifies a significant portion of patients susceptible to undertreatment (high-risk patients), in contrast to the multigene assays that often strive to mitigate overtreatment. The compatibility of our methodology with standard histology using traditional (non-tissue-microarray) formalin-fixed paraffin-embedded (FFPE) tissue sections could simplify subsequent clinical translation.
Keywords: Breast cancer, multiphoton imaging, tumor-associated collagen signatures, disease-free survival
 Global reach, higher impact
Global reach, higher impact